CAR trials drive leukemia and lymphoma treatment in new direction
Cancer immunology is based upon boosting the body's own immune system to vanquish malignancies. It is among the fastest growing areas of oncology research. Researchers at UC San Diego Moores Cancer Center have launched three clinical trials to test the safety and efficacy of a novel cellular-immunotherapy that uses modified T cells - one of the immune system's primary weapons - to treat three different types of blood cancer that often defy existing therapies.
"Lymphomas and leukemias affect thousands of Americans every year and unfortunately a good number of them die as a direct consequence of the disease progression or toxicity from existing treatments," said Januario E. Castro, MD, professor of medicine at UC San Diego School of Medicine and a blood disease specialist at Moores Cancer Center. "We have made great strides with some blood cancers, notably Hodgkin lymphoma, but others have proved more resistant, with patients exhausting all current standards of care."
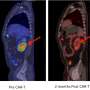
Castro is principal investigator for all three clinical trials, dubbed ZUMA-1, ZUMA-2 and ZUMA-3. The trials utilize the recent development of so-called chimeric antigen receptor (CAR) T cells. These are white blood cells that have been extracted from the patient and genetically modified to contain a gene that produces the CAR protein on the T cell's surface. The engineered CAR-T cells are then reintroduced into the patient with the hope they can bind to and exclusively kill cancer cells that express target proteins, such as CD19, a molecule found on cancerous B cells involved in most lymphomas and leukemias.
The potential treatment is called KTE-C19. The trials are a collaboration between Santa Monica-based Kite Pharma and multiple testing sites, including UC San Diego medical centers in Hillcrest and La Jolla. All three trials are currently recruiting participants.
More information about specific trials and contact instructions are below.
ZUMA-1: Intervention for Refractory Aggressive Non-Hodgkin Lymphoma
Lymphoma is a blood cancer that begins in cells of the lymph system. Most patients with Non-Hodgkin lymphoma (NHL) experience enlargement of the lymph nodes and spleen and frequently bone marrow damage. NHL involves different types of white blood cells (usually B, but also T and NK). It can be indolent (slow-growing) or aggressive; treatment and prognosis depend upon the stage of disease and subtype of NHL.
The ZUMA-1 trial targets patients with refractory, aggressive NHL. When standard therapies have not worked, lymphoma cells remain and disease progression is considered aggressive. KTE-C19 treatment involves three days of chemotherapy, followed by a week-long hospital stay and up to 15 years of follow-up visits. As with all trials, there are specific eligibility criteria to qualify for treatment. For example, ZUMA-1 trial participants must have a confirmed NHL diagnosis, but no anti-cancer therapy for two weeks prior to study enrollment. There must be no central nervous system involvement, no history of hepatitis B or C or HIV infection and no history of another malignancy within the last three years. Full eligibility criteria can be found at https://clinicaltrials.gov/ct2/show/NCT02348216?term=kite&rank=7
ZUMA-2: Intervention for Relapsed/Refractory Mantle Cell Lymphoma
Mantle cell lymphoma (MCL) is a rare, B-cell NHL that most often affects men over the age of 60. It involves tumor cells that originate from the "mantle zone" of the lymph node, an outer ring of small lymphocytes surrounding a germinal center.
It can be slow- or fast-growing, and is usually diagnosed as a late-stage disease that has spread to other regions of the body. The basic trial approach is similar to ZUMA-1: three days of chemotherapy using KTE-C19 for qualified participants, followed by a week of hospitalization and long-term, regular check-ups to monitor disease response and remission.
Eligibility criteria for ZUMA-2 include an MCL diagnosis and many of the same requirements as ZUMA-1. Full criteria can be found at https://clinicaltrials.gov/ct2/show/NCT02601313?term=kite&rank=6
ZUMA-3: Intervention for Relapsed/Refractory B-precursor Acute Lymphoblastic Leukemia
B-precursor Acute Lymphoblastic Leukemia (ALL) is an aggressive form of leukemia characterized by the overproduction and accumulation of cancerous, immature (dysfunctional) white blood cells. In this case, B cells. It is most common in childhood, with peak incidence at 2 to 5 years of age, although ALL also occurs in old age.
As with other ZUMA clinical trials, the study's primary goal is to assess whether the experimental KTE-C19 therapy is safe and effective. Trial participants will undergo similar genetic treatment of their own blood cells to introduce CAR-T cells and similar periods of chemotherapy, hospitalization and clinical visits.
To qualify for ZUMA-3, patients must have morphological disease in their bone marrow and cannot have undergone certain types of treatment within weeks of enrollment. Other criteria are similar to ZUMA-1 and ZUMA-2. Full eligibility criteria can be found at https://clinicaltrials.gov/ct2/show/NCT02614066?term=kite&rank=5