Microreactor replaces animal testing
Researchers all over Europe are working on alternatives to animal testing that can be used to evaluate the adverse side-effects of medications. But many of these test methods still present problems. A microreactor developed as part of an EU-funded collaborative research project enables cultured liver cells to be used as test samples. Unlike animal testing, this novel method enables the assessment of potentially toxic substances on tissue in real time.
Serious efforts are underway to significantly reduce the number of animal tests carried out for research purposes. The latest EU Cosmetics Regulation, which came into force in 2013, bans the sale of cosmetic products containing ingredients that have been tested on animals. But it is difficult to find alternatives, not only for the cosmetics industry but also in the field of pharmaceutical research. In many cases, there are no other suitable methods of toxicity testing available. Numerous research groups are therefore working on the development of new, viable test formats.
One of the most promising approaches involves the use of cultured liver cells. The liver is the most important organ when it comes to eliminating toxins from the body. It therefore makes sense to use liver cells to test the toxicity of substances. But this isn't as easy as it sounds. It has to be ensured that all cells are equally exposed to the substance being tested. And a further problem in the case of liver cells is that they rarely survive for longer than a few days in a laboratory environment. This makes it virtually impossible to carry out experiments to determine the long-term effect of toxic substances on a living organism.
Watching how liver cells react in real time
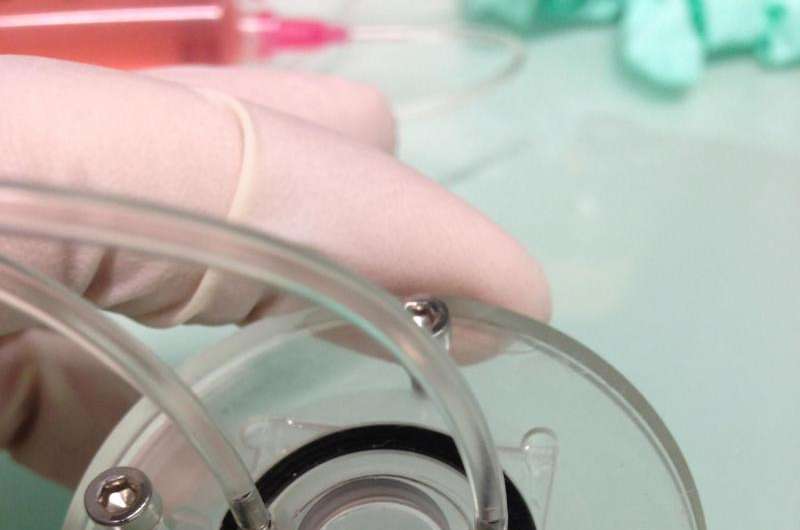
As part of the HeMiBio (Hepatic Microfluidic Bioreactor) project, researchers at the Fraunhofer Institute for Cell Therapy and Immunology IZI in Potsdam in collaboration with partners at the Hebrew University of Jerusalem have developed a microbioreactor in which liver cells can be kept alive and observed for a period of one month. The particular advantage of this device is that it allows researchers to watch how liver cells react to toxic substances directly and in real time. "Up to now, both in animal testing and in conventional lab tests, measurements have usually only been made at the end of the test," says Dr. Claus Duschl, head of the cellular biotechnology department at the IZI. "The procedure consists of administering different doses of an active ingredient and
subsequently analyzing the areas of dead tissue or the dead animal. It is not possible to determine the precise effect of the active ingredient on the cells using this method."
Sensors measure oxygen consumption

The microbioreactor changes all this. Its miniature sensors gather real-time data on the amount of oxygen being taken up by the liver cells at any instant. Oxygen consumption is high when the cell's metabolism is stimulated. If the cell dies, the oxygen consumption falls. By reading this curve, cell biologists are even capable of pinpointing the metabolic processes taking place in a cell at a specific point in time. The HeMiBio project partners make use of this information. When a toxic substance is placed in the microreactor, the sensors record a detailed picture of the changing levels of oxygen consumption. These measurements make it possible to precisely identify the stages of the metabolic process that are affected or halted by the active ingredient. "While working on this project with cell biologists at the Hebrew University of Jerusalem, we have been able to verify various hypotheses by selectively replacing specific metabolic products whose synthesis had been blocked by the toxic substance," Duschl explains. "As we had surmised, the metabolic process then continued unaffected to the next stage."
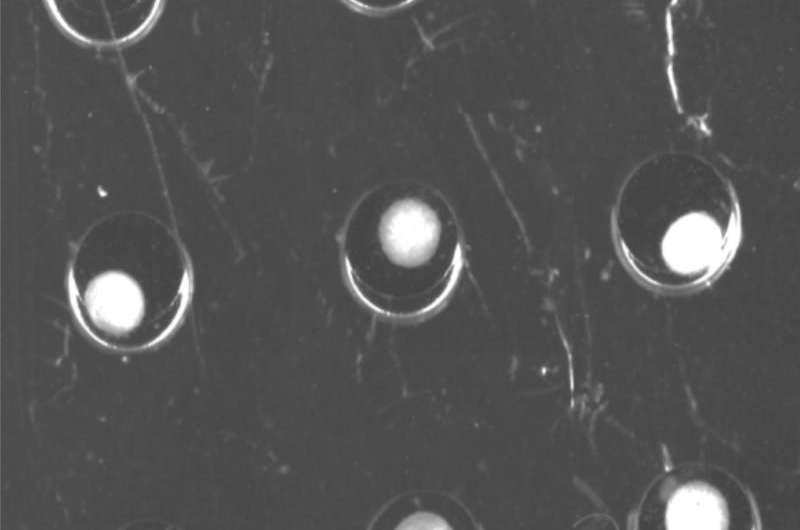
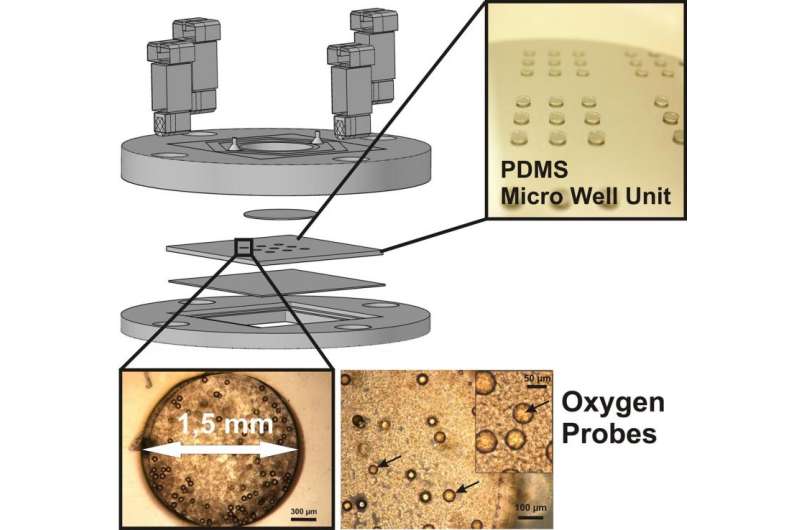
One of the tasks of Duschl's team was to design a suitable reactor vessel containing numerous microfluidic channels, in collaboration with their partners in Israel. One of the main challenges was to ensure that all cells were equally supplied with the growth medium, so they are evenly distributed and do not form clumps. But this fine distribution was a source of other problems, for the wider the distance between the cells, the weaker the signals captured by the sensors. "What we needed was a sensor technology capable of dealing with a high concentration of cells, without the risk of interference effects that would falsify the test results." The IZI project team came up with the idea of using tiny polymer particles containing a luminescent dye. This dye emits a phosphorescent glow when exposed to a monochromatic LED light source, which excites individual electrons and raises them to a higher energy level. The electrons return to their original energy level within a fraction of a second, and the remaining energy is emitted in the form of phosphorescent light. The time required by the electrons to lower their energy level is directly related to the concentration of oxygen in the surrounding air. "In other words, the time this takes indicates the presence of metabolic activity, and can be used to measure the effect of the toxic substance." This is an important factor because it enables scientists to gain a better understanding of the way specific categories of substances affect the human organism and understand why some of them are toxic while others have a therapeutic effect.
Simulating metabolic processes

The participants in the project have proved that the microreactor functions as intended. But they still have plenty more work to do. Because many different types of cells are active in the liver, the researchers now want to populate the reactor with various combinations of cells. "In this way, we will be able to improve our ability to simulate metabolic processes," says Duschl. Eventually the researchers hope to be able to combine tissue samples from different organs in a single reactor. "But we still have a long way to go," says Duschl.