Scientists find a method of protection against Alzheimer's disease
A joint study on the Lynx1 protein has revealed that the protein competes with human nicotinic acetylcholine receptors for binding to amyloids, which are the main cause of Alzheimer's development. In the future, the protein could be used in the development of new therapies or future combinations for the treatment of the disease. The results of the experiment were published in the Neurobiology of Aging journal.
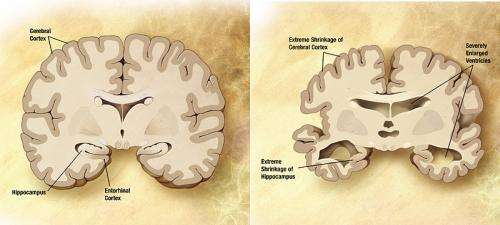
The scientists focused on Alzheimer's disease, a neurodegenerative disease that is accompanied by memory loss, speech disorders, and disorders in spatial orientation and cognitive functions. As has already been established, one of the features of this disease is the accumulation of amyloid, i.e. peptides that form plaques in brain tissues. These plaques create complexes with nicotinic acetylcholine receptors, which leads to the disruption of cognitive processes. The current knowledge base for this disease is still incapable of establishing a clear picture of the mechanisms of the disease. However, a team of scientists consisting of researchers from the Institute of Bioorganic Chemistry of the Russian Academy of Sciences (IBCh RAS) has recently taken a groundbreaking step toward understanding its nature. Their research has revealed the interaction mechanism between the Lynx1 protein, which is responsible for the neuroplasticity of the brain, and the nicotinic acetylcholine receptor, which ensures the transmission of nerve impulses, and the role they play in the development of Alzheimer's disease.
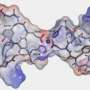
"We know that the Aβ1-42 peptide plays an important role in the development of Alzheimer's disease," says Ekaterina Lyukmanova, Ph.D., one of the authors of the article. "This peptide forms oligomer molecules, which in turn form amyloid plaques. Preliminary research studies on the disease, aimed at fighting this peptide, consist of three ways of solving the problem: preventing the formation of peptides, preventing the creation of oligomers and taking remedial action against the after-effects of the formation of amyloid plaques. We have demonstrated the competition that exists between the Lynx1 human protein, which we can easily obtain using the recombinant production system that is found in E. coli cells, and the Aβ1-42 peptide for binding to the nicotinic acetylcholine receptor."
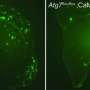
Ekaterina and her colleagues ascertained the ability of Lynx1 to bind to different subunits of the nicotinic acetylcholine receptor that were isolated from different brain regions of rats and humans. The researchers then showed that pre-incubation of brain tissue with a peptide results in a significant decrease in the proportion of the subunit nicotinic receptor extracted using Lynx1. This indicates the competition that exists between the Lynx1 and the amyloid peptide for binding to the nicotinic receptor. In addition, researchers also showed that the Lynx1 reduces the extent of tissue damage that is caused by the Aβ1-42 peptide on cortical neurons of mice by more than 50 percent.
"We have also obtained more evidence for the hypothesis that we made on the relationship between the Lynx1 and Aβ1-42 peptides in Alzheimer's disease," confirms Ekaterina. "The Lynx1 content in the cortical neurons of transgenic mice with pathologies similar to Alzheimer's disease dropped, as compared to that in healthy mice. On the contrary, the peptide Aβ1-42 content increased. This indicates the actual absence of a protective effect for the Lynx1 in transgenic animals."
Understanding the process of the interaction of the Lynx1 with the receptor, as well as a detailed study of the competition that exists between the protein and the amyloid peptide for binding with the receptor, could initiate a new series of experiments in the study of Alzheimer's disease. This mechanism could be instrumental in the development of new treatment methods, or may help in the creation of potentially promising treatment combinations.
More information: Morten S. Thomsen et al. Lynx1 and Aβ1-42 bind competitively to multiple nicotinic acetylcholine receptor subtypes, Neurobiology of Aging (2016). DOI: 10.1016/j.neurobiolaging.2016.06.009