Aging and cancer: An enzyme protects chromosomes from oxidative damage
EPFL scientists have identified a protein that caps chromosomes during cell division and protect them from oxidative damage and shortening, which are associated with aging and cancer.
When cells divide, they pack up all of their genetic material in the tightly wrapped chromosomes. The ends of our chromosomes have a unique structure, named a telomere. Replication of telomeres requires specialized mechanisms, which adult organisms only have in a small number of cells. This means that chromosomes become shorter over time, limiting the lifespan of cells and contributing to aging. Telomeres are also very sensitive to oxidative damage, which affects their ability to replicate. EPFL scientists have now discovered a protein that associates with chromosomes during cell division and protects their tips from oxidative damage. The discovery, published in Cell Reports, could have significant implications for how we could treat cancer and other age-related diseases in the future.
Division, damage, and shortening
Precise transmission of the genome from a cell to its progeny is vital to maintain its characteristics and for the health of the entire organism. Our genome is constantly subjected to damage from environmental factors such as sunlight and oxygen radicals, which are by-products of our normal metabolic functions. As such, oxidative damage is a constant threat to all life on Earth.
Cells have evolved numerous antioxidative defenses, but some parts of the cell, like the chromosome tips, the telomeres, are particularly vulnerable to oxidative damage. Telomeres are sequences of repetitive nucleotides at each end of a chromosome. Their role is to protect that end from damage or from fusing with other chromosomes, which would be catastrophic for the cell. In most adult tissues, every time it divides, its chromosomes shorten a little in length; eventually, the telomeres shorten so much that the end of the chromosome becomes exposed, which causes either the death of the cell or an irreversible block to further divisions. This process is accelerated by oxidative damage. The prevailing theory of aging, as well as cancer, cites a central role for oxidative damage of the telomeres in these processes.
An enzyme that protects telomeres
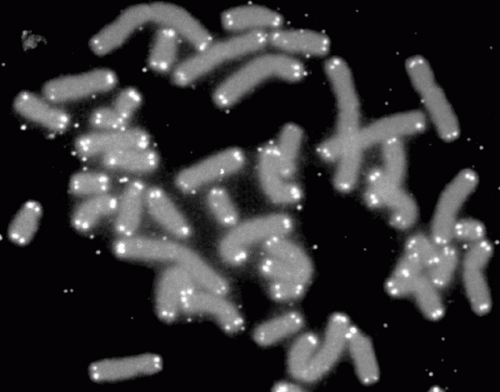
Chromosomes are made up of DNA that is tightly wound up around specialized proteins. The labs of Joachim Lingner and Viesturs Simanis at EPFL analyzed the protein make-up of telomeres across the entire cell cycle to better understand how oxidative damage affects telomeres during division.
The researchers used a number of molecular biology techniques, including a relatively new one called QTIP, which labels various proteins in chromosomes so that researchers can compare and identify quantitative differences between the protein composition of telomeres in various phases of the life cycle.
The study identified an enzyme called Peroxiredoxin 1 (PRDX1). It functions as an antioxidant enzyme, meaning that it is used by cells to mitigate the effects of oxidative damage.
Using QTIP, the researchers found large amounts of PRDX1 on telomeres during two phases of the cell cycle: the phase when the cells synthesizes new DNA and duplicates its genetic material (S-phase), and during the immediately following phase (G2), when the cell grows in size just before it begins dividing.
Using genetic techniques, the scientist removed PRDX1 from the cells, and found that the telomeres were even more susceptible to oxidative damage. This means that PRDX1 plays an antioxidative role that protects telomeres.
In addition, the researchers were able to shed some light onto how oxidative damage affects telomeres. When they incorporated an oxidatively damaged nucleotide into telomeres, they found that the chromosome stopped growing. The reason is that the enzyme called telomerase that builds chromosomes by elongating them abruptly abandoned the process when it encountered the damaged nucleotide. As cancer cells require telomerase for survival this finding may open up novel avenues for attacking this enzyme in cancer.
"Our study links oxidative damage and telomeres, both of which have been previously linked to aging and cancer," says Joachim Lingner. In addition to these, oxidative damage of telomeres is also connected to cardiac failure and muscular dystrophy. Having identified PRDX1, Lingner's lab will now try to determine if there are other antioxidant enzymes that can protect telomeres: "We expect that further studies of this problem will provide insights that help us understand mechanisms of cancer development, aging and inherited disease."
More information: Eric Aeby, Wareed Ahmed, Sophie Redon, Viesturs Simanis, Joachim Lingner. Peroxiredoxin 1 protects telomeres from oxidative damage and preserves telomeric DNA for extension by telomerase. Cell Reports 17, 1-8. DOI: 10.1016/j.celrep.2016.11.071