New gene therapy for pseudarthrosis trialed at Kazan University

A team headed by Professor Albert Rizvanov, director of the Gene and Cell Technologies Open Lab, has created a gene therapy drug that encodes growth factors for the stimulation of blood vessel and bone formation. The combination was highly effective in a patient admitted to the Republican Clinical Hospital in Kazan, Russia. The treatment was approved by the ethical committee, supported by the Ministry of Healthcare of Tatarstan and published in BioNanoScience.
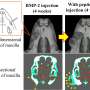
Professor Rizvanov says, "We combined a demineralized bone transplant with recombinant genetic material, which carries genes for vascular endothelial growth factor, to stimulate new blood vessel growth (angiogenesis), and bone morphogenetic protein to stimulate bone growth (osteogenesis). Thus, the survival of transplant and bone tissue formation was achieved at the desired location. We were able to translate our basic and pre-clinical research and are the first to document the efficacy of such therapy in a real clinical case of pseudarthrosis."
Standard methods of bone regeneration use a demineralized bone matrix. Cells and minerals are removed from animal bone tissue, and only the matrix is left—a "bone-like" sponge that can be used to fill in defects in the bone to stimulate the formation of new bone. However, current procedures often result in complications, such as resorption of the transplant. Problems include poor blood supply and low levels of pro-osteogenic growth factors.
The new therapy, a combination of demineralized bone with gene therapy, is a promising solution for complications like pseudarthrosis and other bone defects and fracture treatments. The researchers now plan to offer treatments at the Kazan University Clinic as a part of a new clinical trial program at the strategic academic unit Translational 7P Medicine for biomedical and translational research.
More information: Ruslan Masgutov et al, Use of Gene-Activated Demineralized Bone Allograft in the Therapy of Ulnar Pseudarthrosis. Case Report, BioNanoScience (2016). DOI: 10.1007/s12668-016-0325-7