Mysteries of pregnancy revealed in 3-D imaging by UCSF research team

A team of University of California, San Francisco scientists has turned the mysteries of embryonic development into colorful Disney-like videos, portraying for the first time in detail the precarious journey of the earliest days of life.
Their videos reveal the long-hidden choreography between a mouse embryo and the mother's uterus as it prepares for pregnancy, and their 3-D imaging technique could lead to improved treatments for women, showing what's needed for a successful pregnancy, researcher Ripla Arora and a team from UCSF wrote in the latest issue of the journal Development.
"You need a 'conversation' between the embryo and the mother" as the newly conceived life seeks a secure spot to grow, said Arora, a postdoctoral researcher in the laboratory of co-author Diana Laird, an associate professor of obstetrics, gynecology and reproductive sciences at UCSF.
By understanding these dynamic processes, it becomes easier to identify how and why things go wrong, which could provide a deeper understanding of infertility or miscarriage.
For instance, the technique could help explain why pregnancies fail because of certain genetic mutations, exposure to environmental toxins in plastics and common household products, or even metabolic disease and obesity.
"We hope to finally understand aspects of early pregnancy that remained a mystery," said Laird, "such as how the embryo finds its home within the uterus and what factors are needed for it to implant successfully."
Their 3-D imaging technique has been used in other fields of medicine, such as viewing organs, but until now, it had never been used to view a wandering embryo inside a quickly changing uterus.
The early days of conception are a so-called "black box" in reproductive medicine because two-dimensional modeling doesn't fully capture the complex process.
While scientists have deeply studied the early development of the tiny embryo itself, they have only limited understanding of the much larger environment in a uterus that embryos must navigate.
The changing landscape of the uterus is unexplored terrain. We don't know why it is sometimes welcoming and at other times formidable. As a result, in vitro fertilization doctors can't explain why some implanted embryos flourish while others don't - posing a heartbreaking and expensive experience for couples.
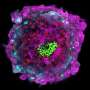
The UCSF team's footage - computationally highlighted, with bright colors - shows a tiny mouse embryo, colored red, implanted in a cozy pocket of its mother's uterus, colored gray, surrounded by multihued glands.
It visualizes for the first time how the structure of the animal's uterus changes as the moment of embryonic attachment comes closer.
The uterine lining becomes folded, like a mountain range, creating little pockets.
The embryo lands in one of these pockets, perhaps preparing itself to attach.
As it attaches, the ductwork of the uterine glands - which provide nourishment - change shape and direction, pointing toward the newly implanted embryo like flowers facing the sun.
Timing is everything. "The uterus is only receptive to attachment of the embryo for a brief time," said Laird.
The researchers suspect these changes are key to the uterus's ability to accept and nourish an embryo through its first weeks.
Visualizing this process is a needle-in-a-haystack challenge. A mouse embryo is roughly 100 microns in size - about the diameter of a human hair. The uterine horn, where it implants, is about 50 times larger.
Two state-of-the-art tools made the discovery possible: high-end microscopes and specialized software to render images of biological tissues.
"This has been the culmination of advanced microscopy techniques and computer science," said Laird.
The beauty of the 3-D model, said Arora, is that it offers a close-up view of the tiny embryo and, by zooming out and rotating, also shows the entire larger uterine structure.
It is unethical to use this same approach to study human pregnancies, because it is invasive and destructive.
But the team's analyses of human uteri, discarded after hysterectomies, shows we share similarities and differences with mice. While the geometry of a human uterus seems less complex, the density and organization of our uterine glands are more complex.
The researchers hope future experiments will reveal whether the human uterus also forms pockets and reorients the glands.
"Being able to do these experiments in mice," said Laird, "will help us understand what makes pregnancy a success in humans, as well."
©2016 The Mercury News (San Jose, Calif.)
Distributed by Tribune Content Agency, LLC.