Exploiting acidic tumor microenvironment for the development of novel cancer nano-theranostics
Cancer is one of leading causes of human mortality around the world. The current mainstream cancer treatment modalities (e.g. surgery, chemotherapy and radiotherapy) only show limited treatment outcomes, partly owing to the complexities and heterogeneity of tumor biology. In recent decades, with the rapid advance of nanotechnology, nanomedicine has attracted increasing attention as promising for personalized medicine to enable more efficient and reliable cancer diagnosis and treatment.
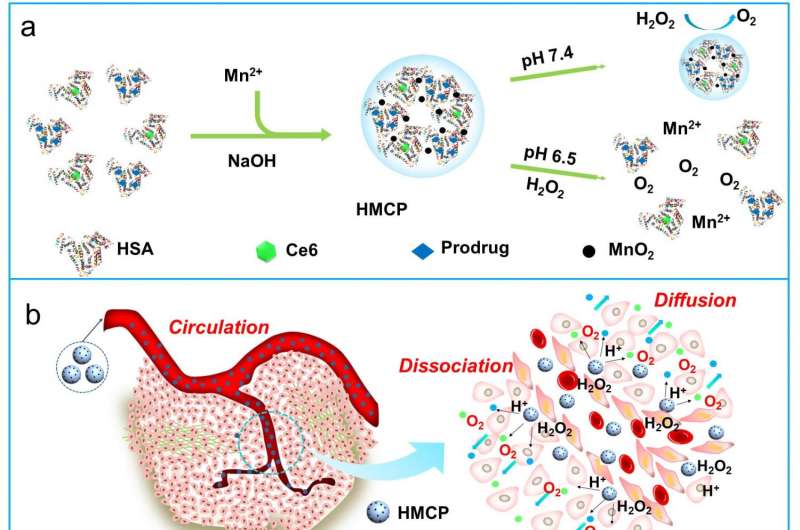
Unlike normal cells energized via oxidative phosphorylation, tumor cells utilize the energy produced from oxygen-independent glycolysis for survival by adapting to insufficient tumor oxygen supply resulting from the heterogeneously distributed tumor vasculatures (also known as the Warburg effect). Via such oncogenic metabolism, tumor cells would produce a large amount of lactate along with excess protons and carbon dioxide, which collectively contribute to enhanced acidification of the extracellular TME with pH, often in the range of 6.5 to 6.8, leading to increased tumor metastasis and treatment resistance.
With rapid advances in nanotechnology, several catalogs of nanomaterials have been widely explored for the design of cancer-targeted nano-theranostics. In a new overview published in the Beijing-based National Science Review, co-authors Liangzhu Feng, Ziliang Dong, Danlei Tao, Yicheng Zhang and Zhuang Liu at the Institute of Functional Nano & Soft Materials (FUNSOM), Soochow University in Suzhou, China present new developments in the design of novel multifunctional nano-theranostics for precision cancer nanomedicine by targeting the acidic TME and outline the potential development directions of future acidic tumor microenvironment-responsive nano-theranostics.
"Various types of pH-responsive nanoprobes have been developed to enable great signal amplification under slightly reduced pH within solid tumors. By taking the acidic TME as the target, smart imaging nanoprobes with excellent pH-responsive signal amplification would be promising to enable more sensitive and accurate tumor diagnosis," they state in the published study.
"As far as nano-therapeutics are concerned, it has been found that the acidic TME responsive surface charge reverse, PEG corona detachment and size shrinkage (or decomposition) of nanoparticles would facilitate the efficient tumor accumulation, intra-tumoral diffusion and tumor cellular uptake of therapeutics, leading to significantly improved cancer treatment. Therefore, the rational development of novel cancer-targeted nano-theranostics with sequential patterns of size switch from large to small, and surface charge reverse from neutral or slightly negative to positive within the tumor, would be more preferred for efficient tumor-targeted drug delivery."
The scientists also write, "For the translation of those interesting smart pH-responsive nano-therapeutics from bench to bedside, the formulation of those nanoscale systems should be relatively simple, reliable and with great biocompatibility, since many of those currently developed nano-theranostics were may be too complicated for clinical translation."
More information: Liangzhu Feng et al, The acidic tumor microenvironment: a target for smart cancer nano-theranostics, National Science Review (2017). DOI: 10.1093/nsr/nwx062