Hypothalamus neurons modulate pancreatic insulin amounts
The brain is key in the regulation of appetite, body weight and metabolism. Specifically, there is a small group of POMC neurons in the hypothalamus that detect and integrate signals related to the body's energy state and activate the appropriate physiological responses. These neurons are sensitive to fluctuations in nutrients such as glucose, fatty acids and amino acids.
Now, a research project co-chaired by Marc Claret and Antonio Zorzano, both members of the CIBERDEM network, reveals the connection between POMC neurons in the hypothalamus and the release of insulin by the pancreas, and describes new molecular mechanisms involved in this connection. The study has been published in Cell Metabolism.
The connection between hypothalamus and pancreas
POMC neurons detect changes in nutrient availability, but the molecular mechanisms involved are not known in detail. Changes in the shape of mitochondria, known as mitochondrial dynamics, serve as a mechanism of energy adaptation in changing metabolic conditions to adjust the needs of cells.
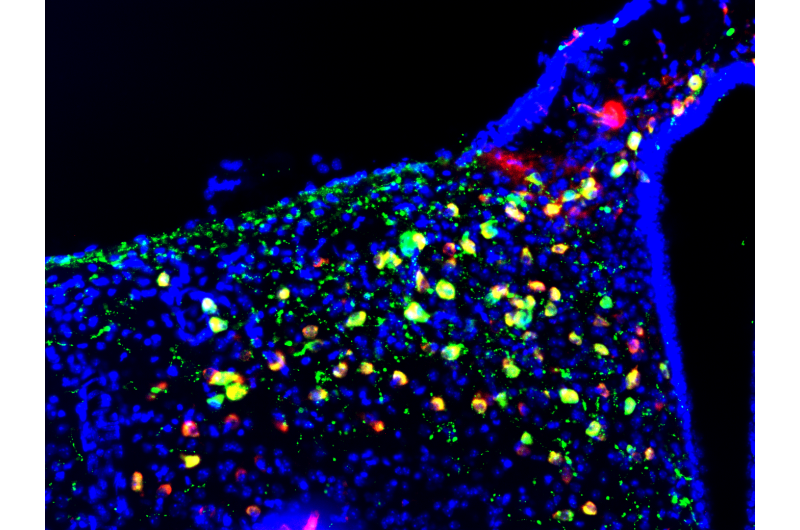
To determine whether defects in the mitochondrial dynamics in the nuclei of POMC neurons could cause alterations in metabolism, researchers removed a mitochondrial protein, Mitofusin 1, in mice.
First, the scientists observed that these mice had altered detection of glucose levels and adaptation between the fasting and fed states. Second, they found that these defects led to disturbances in glucose metabolism caused by reduced secretion of insulin.
"It was surprising to discover that these neurons are involved not only in the control of intake, which was already known, but also in the control of the amount of insulin secreted by the beta cells of the pancreas," explains Zorzano, Head of the Laboratory of Complex Metabolic Diseases and Mitochondria at IRB Barcelona.
Scientists observed for the first time that this communication between the hypothalamus and the pancreas depends on the activity of the protein Mitofusin 1; they are beginning to understand some molecular details of this connection.
They describe that the alterations are due to a disproportionate, though transitory, increase in the production of radical oxygen species (ROS) in the hypothalamus. When the levels of ROS in the hypothalamus are restored in the laboratory, the pancreas secretes the correct levels of insulin.
Obesity and diabetes
Marc Claret, head of the Neuronal Metabolism Control Group at IDIBAPS, says, "Our results also suggest pathological implications of this animal model, since a diet rich in fats makes these mice more susceptible to developing diabetes."
Insulin segregation is a major phenomenon in relation to diabetes. Type 2 diabetes patients, who represent the 85 percent of people with diabetes, have fewer beta cells and reduced ability to secrete insulin in response to glucose.
"Understanding the mechanisms involved in regulating insulin is important, and helps us to better understand the pathophysiology of diabetes," says Claret. "Much research needs to be done to apply these findings, given that we are talking about neural mechanisms of complex intervention."