July 26, 2017 report
Laser used to reawaken lost memories in mice with Alzheimer's disease
(Medical Xpress)—A team of researchers at Columbia University has found that applying a laser to the part of a mouse brain used for memory storage caused the mice to recall memories lost due to a mouse version of Alzheimer's disease. In their paper published in the journal Hippocampus, the team describes their experiments with mice and what their results might mean for human patients.
For many years, medical scientists have believed that when people develop Alzheimer's disease, they lose memories because neurons that encode those memories are destroyed by clumps of protein called beta-amyloid. For this reason, they believed that once lost, such memories can never be restored. But in this new effort, the researchers found that restoring function to neurons known to hold memories of a particular event caused mice to recover a memory that had been lost. This, of course, suggests that Alzheimer's disease does not necessarily destroy memories, but only destroys the mechanism that allows them to be accessed.
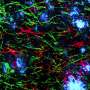
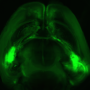
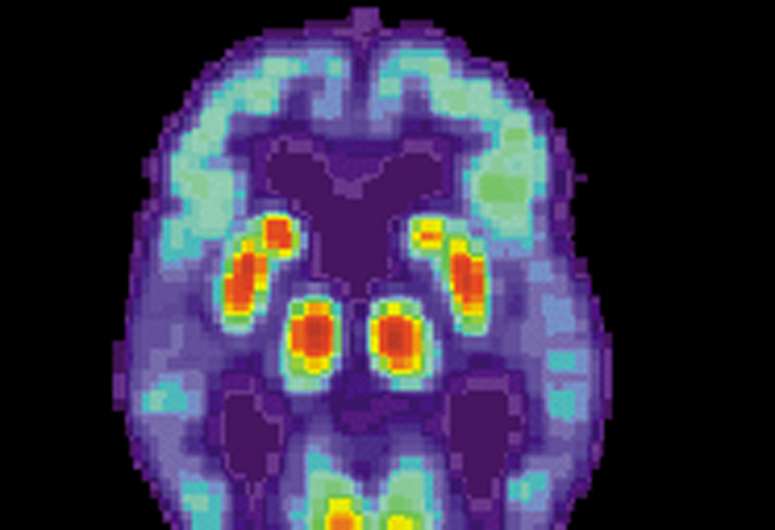
In their experiments, a group of mice was genetically engineered to have neurons that emit a yellow glow when activated as part of memory storage, and another group of neurons to emit red light when activated as part of memory retrieval. The mice were then taught to fear the smell of lemon by shocking them with a small amount of electricity. Later, some of the mice that had developed a mouse model of Alzheimer's disease were tested against a control group—those with the disease had forgotten their fear of the smell of lemon while the control group did not. Both groups were then placed in an MRI machine to observe related activity in their brains, and as expected, those mice with Alzheimer's disease had little activity in the part of the brain that had previously held the fear memories, while the control group had normal activity. In some cases, the team reports, red cells were being activated, indicating that the sick mice were actually experiencing incorrect memories—a common symptom in humans, as well.
In the final part of the study, the researchers pointed a laser at the cells that had previously held the fear memory, and because they had been optogenetically engineered, the neurons were effectively reactivated. The researchers believe the procedure allowed the mice to regain memories associated with the fear of lemon scent.
More information: Jennifer N. Perusini et al. Optogenetic stimulation of dentate gyrus engrams restores memory in Alzheimer's disease mice, Hippocampus (2017). DOI: 10.1002/hipo.22756
Abstract
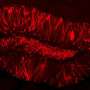
Alzheimer's disease (AD) is a prevalent neurodegenerative disorder characterized by amyloid-beta (Aβ) plaques and tau neurofibrillary tangles. APPswe/PS1dE9 (APP/PS1) mice have been developed as an AD model and are characterized by plaque formation at 4–6 months of age. Here, we sought to better understand AD-related cognitive decline by characterizing various types of memory. In order to better understand how memory declines with AD, APP/PS1 mice were bred with ArcCreERT2 mice. In this line, neural ensembles activated during memory encoding can be indelibly tagged and directly compared with neural ensembles activated during memory retrieval (i.e., memory traces/engrams). We first administered a battery of tests examining depressive- and anxiety-like behaviors, as well as spatial, social, and cognitive memory to APP/PS1 × ArcCreERT2 × channelrhodopsin (ChR2)-enhanced yellow fluorescent protein (EYFP) mice. Dentate gyrus (DG) neural ensembles were then optogenetically stimulated in these mice to improve memory impairment. AD mice had the most extensive differences in fear memory, as assessed by contextual fear conditioning (CFC), which was accompanied by impaired DG memory traces. Optogenetic stimulation of DG neural ensembles representing a CFC memory increased memory retrieval in the appropriate context in AD mice when compared with control (Ctrl) mice. Moreover, optogenetic stimulation facilitated reactivation of the neural ensembles that were previously activated during memory encoding. These data suggest that activating previously learned DG memory traces can rescue cognitive impairments and point to DG manipulation as a potential target to treat memory loss commonly seen in AD.
© 2017 Medical Xpress