September 6, 2017 report
Compound found to improve brain structure development of mice with Down syndrome

(Medical Xpress)—A team with members from several institutions in Japan has found a compound that reduces the impact of Down syndrome in mice. In their paper published in Proceedings of the National Academy of Sciences, the team describes their search for a neural improvement compound and report on one that improved brain structure development in mice with Down syndrome.
Down syndrome is the most common form of chromosomal abnormality occurring in humans, impacting approximately one of every 1000 people. Among other things, it is characterized by reduced brain function. It happens when a person has an extra copy of chromosome 21, which results in problems with nerve development. In this new effort, the researchers began their work by looking for a compound that could cause stem cells to grow into nerve cells. After looking at 717 possibilities, the team came up with one they call altered generation of neurons (ALGERNON). As part of their research, they looked at how the compound might impact a mouse embryo known to have Down syndrome.
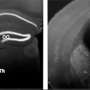
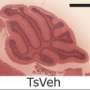
The team studied the impact of the compound by adding it to the feed of a pregnant mouse carrying a Down embryo for a period of five days and then studying its impact on the fetal mouse brain as it developed thereafter. They found that administration of the compound caused the cerebral cortex to develop normally (in mice with Down, the structure is thinner than normal). Even better, they found that the mice behaved normally after birth on into adulthood—they did just as well at solving a maze as unafflicted mice.
The team next tried adding the compound to human stem cells from people with Down syndrome in a petri dish and found that the cells grew normally—this finding, of course, offers the possibility that the compound might also prevent brain malformation for humans, as well. But, the team notes, testing it with human embryos is still a long way off. It is still not known what other impacts the drug might have on development—a lot more testing will have to be done first. The team notes that there might also be some social issues to resolve, because in administering the drug to a pregnant woman, doctors would in effect be altering physical characteristics passed down to offspring from parents.
More information: Akiko Nakano-Kobayashi et al. Prenatal neurogenesis induction therapy normalizes brain structure and function in Down syndrome mice, Proceedings of the National Academy of Sciences (2017). DOI: 10.1073/pnas.1704143114
Abstract
Down syndrome (DS) caused by trisomy of chromosome 21 is the most common genetic cause of intellectual disability. Although the prenatal diagnosis of DS has become feasible, there are no therapies available for the rescue of DS-related neurocognitive impairment. A growth inducer newly identified in our screen of neural stem cells (NSCs) has potent inhibitory activity against dual-specificity tyrosine phosphorylation-regulated kinase 1A (DYRK1A) and was found to rescue proliferative deficits in Ts65Dn-derived neurospheres and human NSCs derived from individuals with DS. The oral administration of this compound, named ALGERNON (altered generation of neurons), restored NSC proliferation in murine models of DS and increased the number of newborn neurons. Moreover, administration of ALGERNON to pregnant dams rescued aberrant cortical formation in DS mouse embryos and prevented the development of abnormal behaviors in DS offspring. These data suggest that the neurogenic phenotype of DS can be prevented by ALGERNON prenatal therapy.
© 2017 Medical Xpress