New health guidance on breast cancer
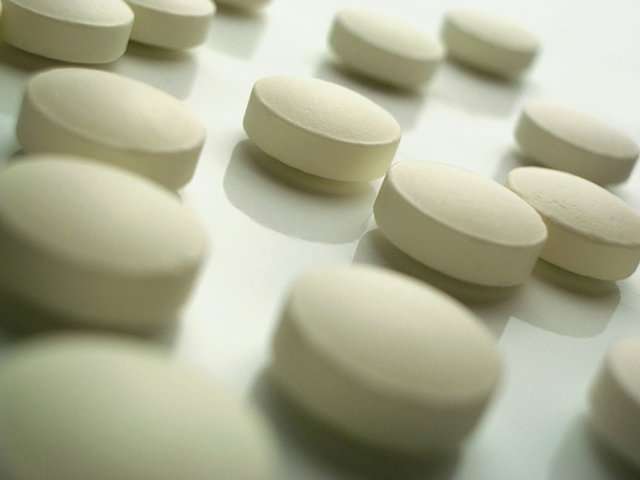
In their draft guidance, the National Institute for Health and Care Excellence (NICE) said that postmenopausal women with a family history of breast cancer should now be offered the drug anastrozole, and not tamoxifen, to help prevent the onset of the disease.
Anastrozole works by preventing the body from making oestrogen - a hormone that fuels many breast cancers.
The IBIS-II trial, led by QMUL's Wolfson Institute of Preventive Medicine, looked at almost 4,000 postmenopausal women at high risk of breast cancer with half being given anastrozole daily and half given a placebo.
The researchers found that participants being treated with anastrozole were 53 per cent less likely to develop breast cancer than those on placebo, and that anastrozole was more effective than previous drugs such as tamoxifen, with fewer side effects.
Based on this research, NICE has now recommended that the breast cancer drug, which costs four pence a day, is offered for five years to women at high risk of breast cancer in England, unless they have severe osteoporosis. Clinicians should also consider offering it to women at moderately increased risk of breast cancer.
Professor Jack Cuzick from QMUL said: "This is an incredibly important development in breast cancer prevention. Since our discovery that anastrozole is more effective and has fewer side effects than tamoxifen, we have known that it should be the drug of choice for reducing breast cancer risk in postmenopausal women who are predisposed to developing the disease. Tamoxifen remains the drug of choice for premenopausal women.
"We are delighted that NICE have now added anastrozole to their recommended drugs for this group of women. By including this drug in their clinical guidelines, this important advancement in preventive medicine will provide greater protection from breast cancer for postmenopausal women at high risk of breast cancer."
Professor Mark Baker, director of the centre for guidelines at NICE, said: "Our draft guideline update recommends the use of anastrozole as a first line treatment for postmenopausal women at high or medium risk of breast cancer who don't have osteoporosis.
"The evidence examined by the committee suggests anastrozole will not only reduce the number of breast cancer cases in these women compared to tamoxifen, but it is also a more cost effective option."
NICE's draft guideline update is out for consultation until 5 January 2017.