Nanosensor that lights up cancer to be tested in surgeries
UT Southwestern Simmons Cancer Center scientists next month will begin testing a digital nanosensor that lights up cancer tissue to see whether it can improve the accuracy of cancer surgeries, thereby reducing cancer recurrence and surgical morbidity.
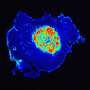
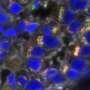
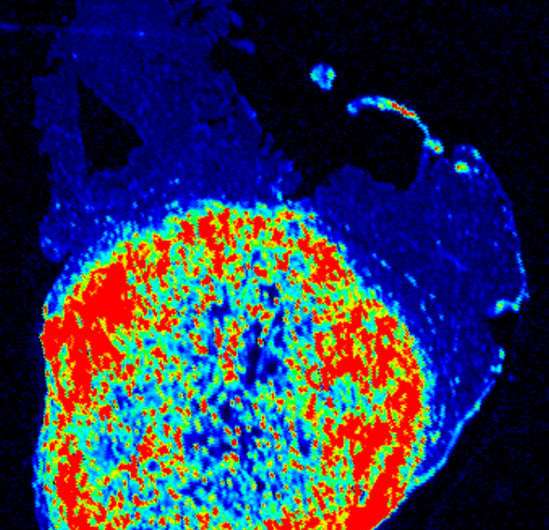
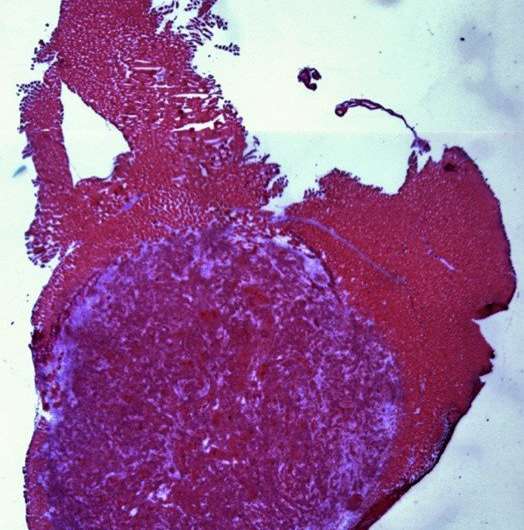
The nanosensor, which works by reacting to low pH, illuminates cancer like a lightbulb, sharply distinguishing cancerous tissue from healthy tissue, and making it easier for surgeons to remove cancer cells while leaving healthy, functional tissue intact.
"We synthesized an imaging probe that stays dark in normal tissues but switches on in solid tumors, behaving like a digital sensor with binary readouts between the two states," said Dr. Jinming Gao, Professor of Pharmacology and Otolaryngology with the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern Medical Center, which is recognizing its 75th anniversary this year. "Cancer is a diverse set of diseases, but it does have some universal features. As solid tumors ramp up, they eat more glucose and secrete lactic acid, so the microenvironment around the cancer cells is acidic."
Dr. Gao and surgeon Dr. Baran Sumer, Associate Professor of Otolaryngology with the Simmons Cancer Center, developed the sensor, and have integrated it with a clinical camera that allows the surgeon to see the fluorescent areas. Their nanosensor not only lights up cancer, but suppresses the signal in normal tissue, leading to a sharp line between cancer and healthy tissue. It is expected to be effective in all solid tumors.
"This new digital nanosensor-guided surgery has several advantages for patients, including more accurate removal of tumors and greater preservation of normal tissue. These advantages can limit the extent of surgery and improve quality of life and, potentially, patient survival," said Dr. Sumer, who leads the Head and Neck Oncology team at the Simmons Cancer Center.
First to be treated with the new technology will be breast cancer patients at the University of Groningen in the Netherlands, which is collaborating in the clinical trials. Patients will be intravenously injected with the nanosensor medication about 12 hours before surgery. Tumors will light up and remain fluorescent for one to two days. Dr. Sumer and Dr. Gao expect to have data on breast cancer and colon cancer surgeries by fall, then hope to move on to Phase II clinical trials in the U.S. and elsewhere.
"What we have is a digital probe that might eventually lead to more exact surgeries so that solid-cancer surgeries would not need to be followed up with radiation or chemotherapy," said Dr. Sumer.

The nanosensor technology and the camera used to see the fluorescent cells are being developed by OncoNano Medicine, Inc., which was the recipient of a Cancer Prevention and Research Institute of Texas product development award to develop this technology. Dr. Gao and Dr. Sumer are paid consultants and scientific co-founders of OncoNano Medicine, Inc. UT Southwestern Medical Center has licensed the technology to OncoNano Medicine and has a financial interest in the research.