Specific microbes in digestive tract can boost success for cancer immunotherapy
In the Jan. 5, 2018 issue of Science, researchers from the University of Chicago Medicine show that specific strains of commensal bacteria - the swarms of microorganisms naturally dwelling in the intestines - can improve the response rate to immunotherapy for patients being treated for advanced melanoma.
Although these immune system boosting drugs have revolutionized treatment of certain cancers, only about 35 percent of melanoma patients who receive them get a significant benefit. In their paper, "The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients," the UChicago researchers demonstrate that several defined bacterial species, including Bifidobacterium longum, Collinsella aerofaciens and Enterococcus faecium, were much more prevalent in the intestines of the 16 patients who responded to treatment (38 percent) than in the 26 patients (62 percent) who did not.
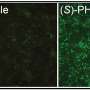
The presence of these specific bacteria in the intestine appears to enhance T-cell infiltration into the tumor microenvironment and augment T-cell killing of cancer cells, increasing the odds of a vigorous and durable response.
The strong correlation between specific gut bacteria and a clinical response to anti-PD-1 immunotherapy suggests "a causal relationship," said study director Thomas Gajewski, MD, PhD, the AbbVie Foundation Professor of Cancer Immunotherapy at the University. "Specific bacteria clearly contribute to improved anti-tumor immunity in patients. The gut microbiota has a more profound effect than we previously imagined."
The current study follows a November 2015 Science paper from Gajewski's laboratory, one of the first to connect the presence of specific intestinal bacteria to greater potency for immunotherapy. These initial papers showed that the composition of the gut microbiome can influence how well these cancer immunotherapies work in mice.
Although the earlier mouse data was striking, "we knew there were multiple barriers that can decrease the odds that immunotherapy will work," Gajewski said. "We initially thought the microbiome was a minor component. But in our current study, these bacteria were a very strong predictor of who would respond."
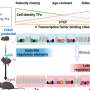
The current paper focuses on humans - 42 patients going through treatment for metastatic melanoma. The researchers collected stool samples from each patient prior to treatment. Thirty-eight patients then received an anti-PD-1 drug, so-called "checkpoint inhibitors such as nivolumab or pembrolizumab. Four more patients received a related drug, anti-CTLA4 (ipilimumab).
The researchers focused on eight bacterial species that were more prevalent in patients who did respond to therapy as well as two species that were more abundant in patients who did not respond. Because of bacterial variations, the team used three different methods to determine the genetic sequences of the microbes.
Patients with a higher ratio of the "beneficial" bacteria to "non-beneficial" bacteria all showed a clinical response, meaning a reduction in tumor size.
The researchers then ran a smaller experiment in reverse. They collected fecal bacteria from three human subjects who responded favorably to treatment, and three who did not respond. They transferred those bacteria into the intestines of germ-free mice. Two weeks later they implanted melanoma cells in the mice.
Two out of three mouse groups given bacteria from human responders had slow-growing tumors and two out of three with fecal material from human non-responders had fast-growing tumors. Mice treated with PD-1-blocking drugs only showed tumor shrinkage if they had microbiota from responding patients.
Two other research teams found similar results but pinpointed different bacteria. A group from the Gustave Roussy Cancer Campus in Villejuif, France, suggested that Akkermansia muciniphila contributed to the antitumor effect of the microbiome. Another team from MD Anderson Cancer Center in Houston, Texas, focused on Faecalibacterium and Clostridiales.
"Our results strongly suggest that the microbiota is a major factor, a gatekeeper for the immune response against a tumor," Gajewski said. "Without microbial support, the immune response just never quite gets going."
"Our results have pushed us in two directions," he added. "We have to start experimenting with probiotics as a way to enhance immunotherapy. We hope to launch a clinical trial using Bifidobacteria in 2018."
They also hope to expand the list of both beneficial and potentially harmful gut bacteria in cancer patients and to identify the mechanisms by which bacteria influence the immune system's response to cancer control.
"Our current results," the authors conclude, "open the avenue for integrating commensal microbial composition along with tumor genomes and germline genetics into a multi-parameter model for maximizing the ability to predict which patients are likely to respond to immunotherapies such as anti-PD-1."
More information: The commensal microbiome is associated with anti–PD-1 efficacy in metastatic melanoma patients, Science 05 Jan 2018: Vol. 359, Issue 6371, pp. 104-108, DOI: 10.1126/science.aao3290 , science.sciencemag.org/content/359/6371/104