Untangling how Epstein-Barr virus infects cells

A team led by scientists at Northwestern Medicine has discovered a new epithelial receptor for Epstein-Barr virus, according to a study published recently in Nature Microbiology.
The study advances the understanding of how Epstein-Barr virus (EBV) and other members of the herpesvirus family enter human cells, said Richard Longnecker, Ph.D., the Dan and Bertha Spear Research Professor and senior author on the paper.
"This could lead to the development of entry-inhibiting drugs that could be used to prevent infection with herpesviruses or treat people with active infections," said Longnecker, also a professor of Microbiology-Immunology and a member of the Robert H. Lurie Comprehensive Cancer Center.
Jia Chen, Ph.D., research assistant professor of Microbiology-Immunology, was first author of the paper, and Xianming Zhang, Ph.D., research assistant professor of Pediatrics and Bethany Perez-White, Ph.D., research assistant professor of Dermatology, were also co-authors.
EBV, which commonly causes mononucleosis, is one of nine types of herpesviruses that can infect humans. Others include herpes simplex virus (HSV), varicella zoster virus (VZV), cytomegalovirus (CMV) and Kaposi sarcoma virus (KSHV).
In humans, EBV infects two types of cells: B cells and epithelial cells; Epithelial cells provide a barrier to that outside world, lining all external surfaces of the human body, while B cells are a type of white blood cell.
Previous studies have described the entry mechanism for EBV into B cells, largely from the collaborative work of the Longnecker laboratory at Northwestern Medicine, the Jardetzky laboratory at Stanford and the Zhou laboratory at the University of California, Los Angeles. Up until now, however, EBV's mechanism of entry into epithelial cells was a mystery.
"The key missing piece was the epithelial receptor," Longnecker said. "Previous studies had suggested the integrins were the entry receptor, but work in our laboratory using CRISPR-Cas9 gene deletion had found integrins were not essential for EBV epithelial cell entry."
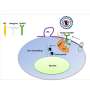
Instead, the Longnecker lab used a novel bioinformatics approach, analyzing gene expression in cells susceptible to EBV infection and cross-referencing it with gene expression in cells resistant to EBV infection. This approach identified EphA2, a protein-encoding gene, as a potential receptor.
Longnecker then used CRISPR to delete EphA2 from epithelial cells, which blocked infection, validating EphA2's role as an entry receptor. Further, EphA2 is often activated in cancers, raising the possibility it may be involved in early cancer development.
While this study doesn't instantly chart the entire mechanism of infection, it provides an important jumping-off point for future work, Longnecker said.
"We will continue to investigate the mechanism of EBV infection in epithelial cells, as well as the potential link of EphA2 signaling in epithelial cancer," Longnecker said.
More information: Jia Chen et al. Ephrin receptor A2 is a functional entry receptor for Epstein–Barr virus, Nature Microbiology (2017). DOI: 10.1038/s41564-017-0081-7