Study reveals the liver's 'weak spot' for hepatitis B virus replication
In a surprising discovery, researchers from the Singapore Immunology Network (SIgN), A*STAR, have found that the liver is the main site of hepatitis B (HBV) replication—not only because it contains material that helps the virus proliferate, but also because most other tissues of the body contain proteins that actively repress HBV replication.
"We've been working on this project for a number of years because HBV is very prevalent in Asia," says Ee Chee Ren, leader of the SIgN team that made the discovery.
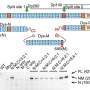
"HBV enters the liver cells through specific receptors and then deposits its genetic material into the nucleus," explains SIgN's Hui Ling Ko, the paper's first author. The viral DNA then hijacks the host cell's machinery to make new copies of HBV. While it's known that liver cells contain factors that facilitate HBV replication, Ko and her team found that the cells also lack functional levels of the proteins Slug and SOX7. These proteins are found in most other human tissues, where they bind to the HBV genome and block the binding of the cells' proteins that initiate and promote viral replication. "Slug and SOX7 are involved in embryonic development and that's when they're most active," says Ko. After development, it is thought that the proteins are no longer required in the liver, and so get 'turned off.'
The team is now using tools including CRISPR-Cas9, the pioneering gene editing technology, to produce cells without any functioning Slug or SOX7 proteins. This nullifies the cells' defenses against HBV infection so that they can be used to test the efficacy of new drugs. "The reason for the very successful development of antiviral drugs for hepatitis C was the availability of robust, cell-based screening assays. Hepatitis B has always been lacking in this area," says Ren. "When we introduce HBV into these Slug-deficient cell lines, they show a large increase in the viral load. This provides us with a dramatically improved platform to create a drug screening assay for HBV."
The SIgN team made another unexpected discovery during their studies: human colon, lung and stomach cells also lack Slug and SOX7. The scientists are now collaborating with clinicians at Singapore General Hospital to launch a thorough investigation into whether these tissues could act as a hidden reservoir of HBV, potentially complicating disease treatment.
More information: Hui Ling Ko et al. Identification of Slug and SOX7 as transcriptional repressors binding to the hepatitis B virus core promoter, Journal of Hepatology (2017). DOI: 10.1016/j.jhep.2017.08.033