Researchers uncover how cancer stem cells drive triple-negative breast cancer
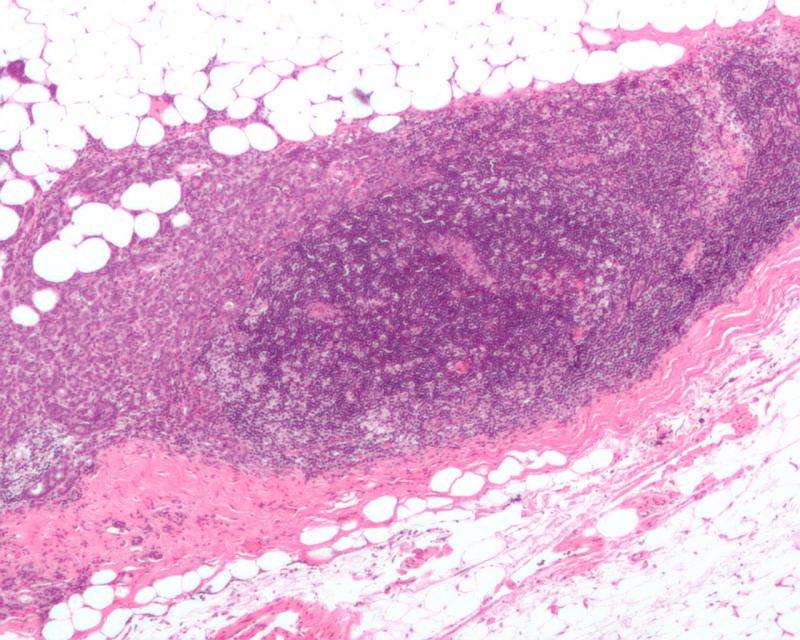
Cleveland Clinic researchers have published findings in Nature Communications on a new stem cell pathway that allows a highly aggressive form of breast cancer - triple-negative breast cancer - to thrive.
Hormone therapy for breast cancer blocks cancer cells from interacting with hormones such as estrogen and progesterone, which fuel the cancer cells to grow and spread. However, triple-negative breast cancer cells lack the receptors needed to bind to these hormones and growth factors. Without such receptors, typical therapy does not work, contributing to poor survival rates for women with this subtype of breast cancer.
Cleveland Clinic Lerner Research Institute researchers believe that an aggressive population of cancer cells, called cancer stem cells, is at the heart of why many cancers, including triple-negative breast cancer, are challenging to treat. Cancer stem cells self-replicate, rapidly grow and spread, and change their phenotype in response to the tumor environment.
The new study, led by Justin Lathia, Ph.D., and Ofer Reizes, Ph.D., identifies a never-before described survival pathway in cancer stem cells that may serve as a potential target for new triple-negative breast cancer therapies.
"Triple-negative breast cancer is resistant to treatment and has a high recurrence rate," Reizes said. "This aggressive subtype accounts for about 15-20 percent of breast cancers. Our findings are at an early stage but we are hopeful that targeting these cancer stem cells will lead to new treatments to allow women to be treated successfully and improve their outcomes."
The team studied a protein called connexin 26 (Cx26), which belongs to the connexin class of proteins. While once believed to suppress tumors, recent research suggests that connexins may actually support tumor progression by aiding in cell-to-cell communication.
The researchers compared healthy breast tissue to triple-negative breast cancer tissue and found that Cx26 is the most highly expressed connexin in diseased tissue vs. normal breast tissue. They also observed that Cx26 levels are higher in cancer stem cells than non-cancer stem cells and that the protein is expressed inside the cell rather than on the cell surface. The researchers also found that Cx26 is linked to two other proteins known to independently promote tumor maintenance and growth.
"Additional research is needed, but this discovery suggests that inhibiting Cx26 and the related pathway may be a promising new strategy for stopping or preventing triple-negative breast cancer stem cells from self-renewing and spreading," Lathia said. "It may also offer a target for diagnostic testing that helps clinicians predict health outcomes and relapse-free survival for patients with a specific cancer type."
More information: Praveena S. Thiagarajan et al, Cx26 drives self-renewal in triple-negative breast cancer via interaction with NANOG and focal adhesion kinase, Nature Communications (2018). DOI: 10.1038/s41467-018-02938-1