Clearer vision of what's inside a tumor and what's going on in there
Researchers at the University of Tübingen have succeeded in combining tumor data from imaging methods and high-throughput technologies with high precision. The goal is to make metabolic processes in tumors visible in their entirety and thus to better understand them. For this purpose, image data from positron emission tomography (PET) and computer tomography (CT) were combined with protein and metabolic data. The research team led by Professor Bernd Pichler from the Werner Siemens Imaging Center at the University of Tübingen published its results in the scientific journal PNAS.
"We shouldn't regard a tumor as a homogenous unit," says Pichler. "Rather, the diseased tissue is often highly heterogeneous." In different parts of a tumor there are differing signaling pathways and receptors and various proteins, which are key for signaling in cells. "Against this backdrop, it is not enough to do a biopsy to understand the tumor in its entirety," he says. In a biopsy, only a tiny sample is usually taken for analysis.
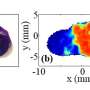
The new method aims to localize the tumor precisely, remove it completely and freeze it for subsequent analysis. Using what's known as multimodal imaging technologies, which combine several methods such as PET-CT and simultaneous measurement, scientists can represent the different types of tissue without penetrating them. "In this way, we gain a precise insight and can also observe the activation of different metabolic pathways within the tumor," says Dr. Marcel Krüger, one of the first authors of the study. "However, we still know very little about the molecular causes of tissue alterations and their effects on the further course of tumor growth. The high-throughput processes can provide better information."
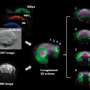
In methods such as proteome and metabolome analysis, the entire protein stock of a sample, the proteome, or all available metabolic products, the metabolome, are systematically recorded. "This provides us with a detailed insight into the molecular processes in the tissue samples, but without any connection to spatial information," adds Dr. Jonathan Disselhorst, the first author. This is how the idea was born to combine the advantages of both methods.
Time and space are recorded during sampling
"The prerequisite for this is that we are able to take the samples for proteome and metabolome analysis with spatial precision on the basis of the images," explains Bernd Pichler. The time factor also plays a decisive role. After surgical removal, the metabolic processes in a tumor change in a very short period of time, so that valuable information about the tumor tissue is lost during a conventional sampling procedure. Rapid freezing captures the metabolism in a snapshot. The scientists were able to demonstrate the effectiveness of the new method in animal experiments.
In order to enable a spatially precise sampling, the tumor areas in the PET and CT data for the examination are defined and transferred to a machine. This in turn exposes the corresponding tissue areas so that they can be removed. "The tissue samples remain frozen throughout the entire procedure and are therefore suitable for subsequent analysis using various high-throughput methods," says Pichler. This enables the researchers to assign the proteome and metabolome data to specific sections of the tumor. The new combination procedure could thus contribute to a better understanding of the course of the disease.
The new method has been established in animal experiments, but the researchers believe it has great potential for clinical application. "For example, it is conceivable to deep-freeze a human tumor immediately after surgical removal and to precisely determine its biological and pathophysiological profile with the new method," explains Pichler. In the medium term, the aim must be to adapt the treatment of patients to the particularities of their disease, i.e. to personalize it. The most comprehensive possible combination of image data and data from protein and metabolism is an important step toward achieving that.
More information: Jonathan A. Disselhorst et al. Linking imaging to omics utilizing image-guided tissue extraction, Proceedings of the National Academy of Sciences (2018). DOI: 10.1073/pnas.1718304115