Biomarker helps identify 'window of opportunity' for cancer chemotherapy timing
Angiogenesis, the formation of new blood vessels, is essential for tumor growth. A new study reported in The American Journal of Pathology describes a vascular stabilization biomarker that can visualize blood vessel activity, thus optimizing the timing of anticancer therapies including anti-angiogenics.
Combination therapy using angiogenesis inhibitors and anticancer drugs can improve drug delivery into tumor tissues and prolong progression-free survival. "Vascular normalization by angiogenesis inhibitors, such as vascular endothelial growth factor (VEGF) signaling inhibitors, is a promising method for improvement of chemotherapy. However, it is unclear how we can recognize the 'window of opportunity' for the tumor vascular normalizing period for effective timing of anticancer drug treatment. Therefore, biomarkers delineating this window are essential," explained Nobuyuki Takakura, MD, PhD, Professor, Department of Signal Transduction, Research Institute for Microbial Diseases, Osaka University, Osaka, Japan.
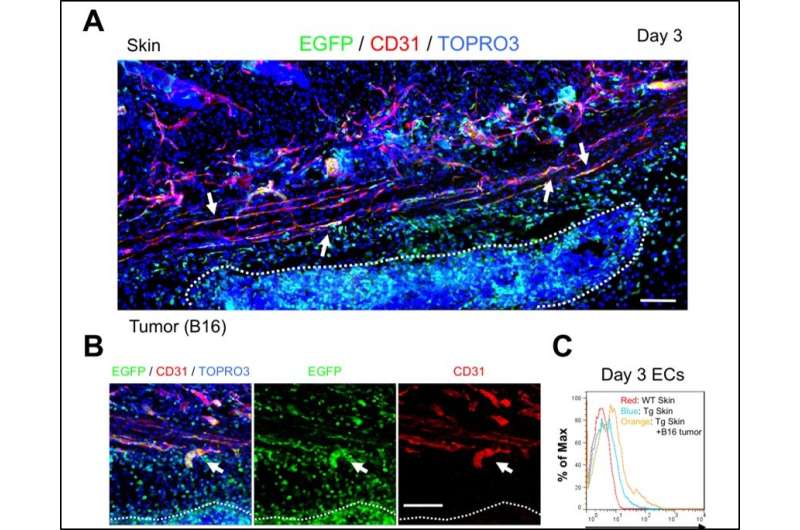
Researchers showed that active proliferating vascular endothelial cells (ECs) in mice could be distinguished from dormant ones. They measured the promoter activity of DNA replication factor partner of Sld5-1 (PSF1; official name GINS1) in ECs using enhanced green fluorescent protein (EGFP) that allows visualization of gene activity as fluorescence.
No EGFP signals were observed in normal adult skin vasculature, which was expected as normal skin ECs are dormant. However, after subcutaneous injection of tumor cells, some ECs in and near the tumor shifted to EGFP-positivity. PSF1 promotor activity was also found to correlate well with tumor cell growth. ECs that were high in EGFP expression were larger and had greater intracellular complexity than cells that were EGFP negative. "Our data showed that PSF1-promotor-EGFP mice may be utilized to visualize proliferating ECs by their EGFP expression," commented Dr. Takakura.
"Experimentation on non-proliferative ECs revealed that these quiet cells strongly expressed VEGFR1 and a cell surface protein CD109. CD109 expression in ECs increased three to five days after injection of bevacizumab into human colorectal adenocarcinoma HT29-bearing mice, coinciding with normalization of tumor vessels. Though on day 5 after bevacizumab injection, functional vessels increased and hypoxic regions significantly decreased, by day 8, hypoxic regions increased again."
These results enabled researchers to successfully distinguish between proangiogenic ECs and quiescent ECs by their PSF1 gene promoter activity, which is associated with DNA replication and rapid proliferation of somatic cells. Therefore, CD109 expression in ECs marked normalized or silenced blood vessels in the tumor vasculature.
"Since CD109 is highly expressed in dormant ECs, we suggest it can be used to detect normalized blood vessels, thus allowing identification of the 'window of opportunity' for optimal delivery of chemotherapeutics," remarked Dr. Takakura.
Though angiogenesis therapy is clinically used to suppress tumor growth, unfortunately, monotherapy using anti-angiogenics such as VEGF signaling inhibitors does not effectively suppress tumor growth in patients. Adding an anti-angiogenic drug can boost an anticancer drug's effectiveness. Basic research indicates that anti-angiogenic therapy allows the blood vessels to return to quiescence and "normalize" so that the anticancer drug can penetrate the tumor more effectively.
More information: American Journal of Pathology (2018). DOI: 10.1016/j.ajpath.2018.01.015