Lung stem cells repair airways after injury
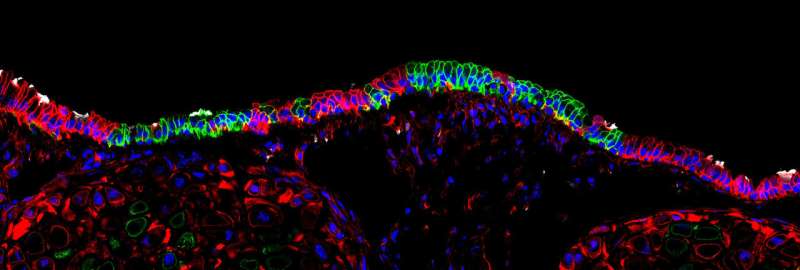
The human airway is a system of branching tubes that connects the nose and mouth with the lungs and allows us to inhale air, extract the vital oxygen, and exhale the waste product carbon dioxide. A layer of epithelial cells helps protect airways from harmful materials in the air we breathe. However, the cells that make up this first line of defense are vulnerable to damage and rely on local stem cells to repair and renew the barrier after injury.
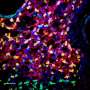
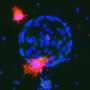
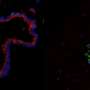
Working in mice, researchers at the University of Iowa have identified a new population of stem cells that appear to be important for regenerating the airway following severe injury. The cells, known as glandular myoepithelial cells (MECs), were found to be surprisingly flexible. The study, which was published April 12 in the journal Cell Stem Cell, found that MECs are able to develop into new replacement cells within their local environment, known as submucosal glands. More surprisingly, the MECs were also shown to be "reserve" stem cells for the lining of the airway, springing into action when the surface of the airway experiences severe damage, and developing into replacement cells there, too.
"We demonstrated that MECs can self-renew and differentiate into seven distinct cell types in the airway," says study co-first author Preston Anderson, a UI undergraduate working in the lab of John Engelhardt, UI professor and DEO of anatomy and cell biology. "No other cell type in the lung has been identified with this much stem cell plasticity."
Because humans have submucosal glands throughout the airway, this specialized niche for MECs may play a significant role in lung regeneration and disease.
The team further investigated the signaling mechanisms controlling the MECs regenerative ability. Focusing on a transcription factor protein called Lef-1 that they had previously shown to be important in glandular development, the researchers found that upregulation of Lef-1 enhances the ability of MECs to migrate, proliferate, and differentiate. Moreover, overexpression of Lef-1 in MECs was sufficient to activate the cells' regenerative response even in the absence of airway injury.
This finding suggests that manipulating Lef-1 signaling in MECs could have major implications for developing practical regenerative medicine approaches to treat airway diseases, such as asthma, chronic obstructive pulmonary disease (COPD), and cystic fibrosis.
"We have identified a potentially important stem cell target and defined a central mechanism that engages stem cell regeneration," says Thomas Lynch, PhD, UI postdoctoral researcher and co-first author of the study. "We hope this new knowledge accelerates the development of regenerative medicines such as gene therapies and pharmaceuticals for lung diseases."
More information: Thomas J. Lynch et al, Submucosal Gland Myoepithelial Cells Are Reserve Stem Cells That Can Regenerate Mouse Tracheal Epithelium, Cell Stem Cell (2018). DOI: 10.1016/j.stem.2018.03.017