Beyond PD-L1: Taking away TIM3 and Tregs stops cancer regrowth after immunotherapy

Radiation treatment can boost the effectiveness of anti-cancer immunotherapy. Still, some patients fail to respond to this combination, and while the combination of radiotherapy and immunotherapy is very good at creating remission, it's not very good at sustaining it. Now a University of Colorado Cancer Center study presented at the American Association for Cancer Research (AACR) Annual Meeting 2018 shows how tumors may learn to overcome this combination treatment: Increased TIM3 and/or increased regulatory T cells (Tregs) within the tumor may help cancers inactivate immune system killer T cells that would otherwise identify and attack the cancer. The findings may help design combination therapies that reduce relapse after immunotherapy.
"Sometimes you give radiation alone and it doesn't work, or you give immunotherapy alone and it doesn't work," says Sana Karam, MD, PhD, investigator at the CU Cancer Center and assistant professor in the CU School of Medicine Department of Radiation Oncology. "But combine the two and—boom!—you get this huge synergy. Unfortunately, the effect is often transient, and we wanted to know why."
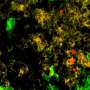
To answer this question, Karam and colleagues including first author Ayman Oweida, PhD, analyzed tumor RNA during "response" and "resistance" phases, allowing them to look for differences in gene activation when radiotherapy/immunotherapy was working and when it eventually stopped working. There was, in fact, a stark difference: In tumors that had become resistant to combination radiotherapy/immunotherapy, the gene TIM3 was dramatically more active.
This makes sense - tumors likely use TIM3 to hide themselves from the immune system.
Here's how it works: Immune system T cells search for and attack foreign cells. But during a pregnancy, for example, there are foreign-like cells that we would rather these T cells not attack. For this reason, T cells come equipped with an "off" switch. T cells coat themselves in a protein called PD-1. And some cells present a partner called PD-L1. When the PD-1 on T cells detects PD-L1 on another cell, the T cells are turned off and do not attack. Many cancer cells express PD-L1 as a way to cloak themselves from the immune system and a successful strategy of anti-cancer immunotherapy has been the use of PD-L1 inhibitors such as pembrolizumab and nivolumab to stop the ability of cancer cells to express PD-L1 and thus hide from immune system T cells.
The interaction between T cells' PD-1 and tumor cells' PD-L1 is called a "checkpoint." TIM3 is an essential link in another checkpoint. The current study implies that when immunotherapy inhibits PD-1/PD-L1 checkpoint, cancers may learn to use the TIM3 checkpoint instead to hide from the immune system.
When Karam and colleagues added an experimental TIM3 inhibitor to radiotherapy/immunotherapy they were able to increase the duration of response in models of head and neck cancer.
"We got a much better response," Karam says. "You go from 30 days to 60 days, but if you watch long enough, you still end up with regrowth. The tumors still return."
The question remained: How were tumor cells evolving to resist therapy, this time in response to anti-PD-L1, anti-TIM3 and radiotherapy? The study shows that the answer may have to do with another of the body's important "off" switches.
"If you have a cold or infection, T cells come in to destroy the bacteria," Karam says. "But if T cells are constantly on, they result in autoimmune disease. Nature is so smart that you have a population of cells that can turn off these T cells. We call these regulatory T cells, or Tregs."
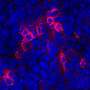
When the group again explored differences between tumors that responded to this three-part therapy and tumors that were resistant, they found that resistant tumors were shot-through with Tregs. In other words, the tumors had again discovered a way to inactivate immune system T cells, this time by the use of Tregs.
But when the group went a step further, testing TIM3 inhibition with PD-L1 inhibition with radiotherapy in the absence of Tregs, "We eradicated the tumor completely," Karam says. Not only did this combination therapy eradicate the existing tumor, but when Karam reintroduced cancer cells to the model, these cells failed to grow new tumors.
TIM3 inhibition has already been hypothesized as an immunotherapy alone or in combination with PD-L1 inhibition, and experimental TIM3 inhibitors already exist. However, no strong strategy exists to rid tumors of Tregs. The group's ongoing work hopes to pick apart the strategies that Tregs use to inactivate T cells.
Intricacies of the science remain. But discovering and inhibiting the mechanisms that tumor cells use to inactivate the immune system, Karam says, "could be a game-changer for clinical practice in the way we manage many cancers."