Researcher: Big data, networks identify cell signaling pathways in lung cancer
A team of scientists led by University of Montana cell biologist Mark Grimes has identified networks inside lung cancer cells that will help understand this cancer and fight it with drug treatments.
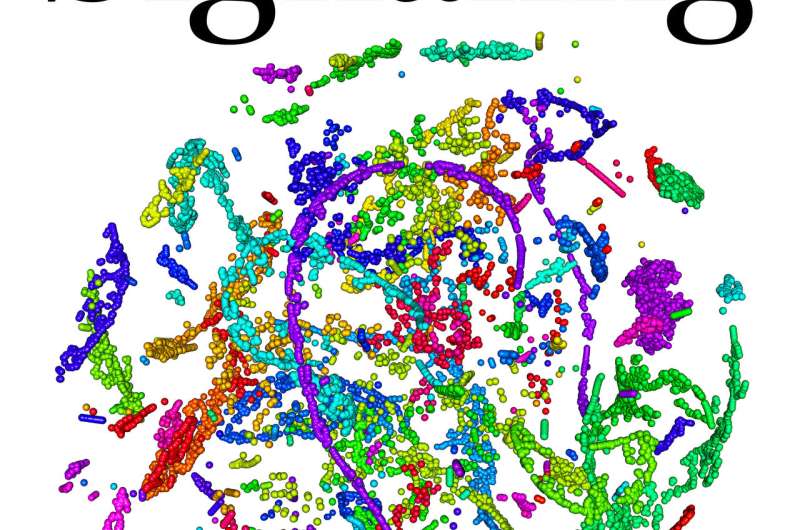
The paper, published as the cover article of the May 22 issue of Science Signaling, sheds light on how a new class of drugs called HSP90 inhibitors—which are currently in clinical trials—works to fight cancer. It is the first large-scale study to analyze three different kinds of protein modifications simultaneously while also employing new methods that involve mathematical and computational approaches and networks to analyze these data.
The paper is titled "Integration of protein phosphorylation, acetylation, and methylation datasets to outline lung cancer signaling networks."
Compared to all other cancers, lung cancer is still the most deadly, despite reductions in recent years due to decreases in cigarette smoking. Lung cancer is expected to kill about 150,000 people in the United States this year, according to the American Cancer Society.
The study began with scientists at Cell Signaling Technology, who collected a large amount of data from lung cancer cells. The approach to analyze the data builds on previous work from the Grimes laboratory.
"Proteins modify each other in different ways inside cells to convey signals," Grimes said. "The signals tell cells to divide, differentiate or die. Cancer is many diseases, and many types of cancer are caused by screwed up cell signaling mechanisms. This study defines these signaling pathways with greater precision and integrates pathways that use different protein modifications."
The study looked at 45 different lung cancer cell types and compared their modified proteins to normal lung tissue. To make sense of the large amount of data, sophisticated pattern recognition techniques—including machine-learning algorithms—were used, and patterns in protein modifications were combined with protein interaction networks to define cell signaling pathways in lung cancer cells.
Grimes' research team includes three UM students and two other UM faculty members: Travis Wheeler from the Department of Computer Science and Ekaterina Smirnova from the Department of Mathematical Sciences. The team also included computational biologist Avi Ma'ayan and his team from Icahn School of Medicine at Mount Sinai, New York; and scientists from Cell Signaling Technology, Danvers, MA led by Michael Comb.
Grimes emphasized that this research is a team effort.
"Progress is made when people with different skills come together to tackle a hard problem," he said. "It was gratifying to involve UM graduate and undergraduate students Lauren Foltz, Jeremiah Gaiser and William Cook, who each contributed uniquely to the project."
Due to the complexity of the information, Grimes offers an analogy to get across the main points of the paper:
"Cells are bags of proteins and other components like DNA. Different components are contained within intracellular compartments walled off by membranes. Imagine the cell as a house, and people within are proteins. Different groups of people spend more time in different places, depending on their jobs in the house. In the cell, proteins modify one another by attaching phosphate, methyl and acetyl groups, which is akin to people attaching different balloons to one another. The balloons attract particular people to come together, depending on which balloon is carried. This sends a signal to the people attracted to the balloon holder to move within the house and engage in some activity. For example, someone hands you a red balloon and you assemble a group of cooks to go to the kitchen and make a meal.
"We can represent the way people interact with one another in the house as a network. Most of the people in the house interact with many other people over the course of a year, so the network of interactions looks like a hairball, which is not very informative. We want to unravel the hairball, picking out pathways through the network that depend on who gets the balloons. This is where mathematical tricks and machine-learning algorithms come into play. These pattern-recognition techniques help us to identify clusters of people who get different balloons—clusters of proteins that become modified—in similar ways. We then look at interactions only among those clusters of people. In other words, the clusters are used to filter the interactions. This cluster-filtered network is a much simpler network, and informs us about signaling pathways specific to lung cancer cells."
Grimes continues, "To take the analogy further, a tumor is like a house that turns itself into a large factory that uses up the community's energy resources in a residential zone of single-family dwellings. Researchers are like detectives who examine the people holding three different types of balloons simultaneously in many different factories (tumor cell types). The detectives use this information to trace pathways of communications that are common when a house breaks the community rules.
"The cluster-filtered network identifies the bad guys and their collaborators so we have a better idea who should be arrested by law enforcement to stop the house from expanding in the wrong neighborhood," Grimes said. "In other words, this study gives us a better idea which proteins to target with drugs to arrest different types of lung cancer."
More information: Integration of protein phosphorylation, acetylation, and methylation data sets to outline lung cancer signaling networks, Sci. Signal. 22 May 2018: Vol. 11, Issue 531, eaaq1087 , DOI: 10.1126/scisignal.aaq1087