Study of first birth cohort to receive HPV vaccine shows the treatment works
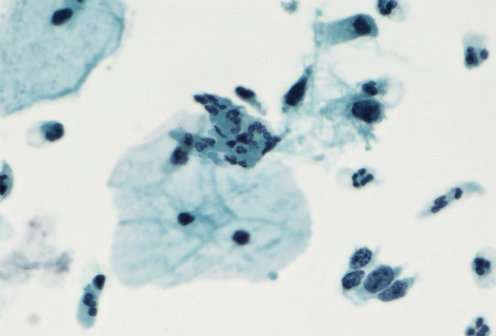
Compared to a birth cohort from 1983, girls in the first birth cohort to receive the HPV vaccine showed a lower degree of dysplasia that could eventually lead to cervical cancer. This is the conclusion of a new study conducted by researchers at the University of Copenhagen, who have conducted the first study of the vaccine's effect on the general population.
The effects of the HPV vaccine, which in 2009 became part of the Danish childhood vaccination programme, have been examined by researchers from the Faculty of Health and Medical Sciences at the University of Copenhagen, and the conclusion is unmistakable: The HPV vaccine works.
The new study, recently published in the scientific International Journal of Cancer, is the first to examine the effect of the vaccine in the population at large, according to Professor Elsebeth Lynge and Ph.D. student Lise Thamsborg from the Department of Public Health, University of Copenhagen.
"It is the first study in the world to test the Gardasil-4 vaccine on a population level. The childhood vaccination programme, which includes the HPV vaccine, is targeted at the entire population. Therefore, it is important to look at the entire population and the effect of the vaccine after the first screening of women aged 23 years," says Professor Elsebeth Lynge, last author of the study.
Reduction in Severe Dysplasia
The researchers have looked at the 1993 birth cohort, which was the first birth cohort to receive the vaccine. They compared it to a 1983 birth cohort that did not receive HPV vaccination. The two birth cohorts of women are comparable and resemble each other as regards level of education and average age of sexual debut, among other things.
The researchers examined the results of the women's first cervical screening tests. The 1993 birth cohort was invited to a screening test in 2016, while women born in 1983 had their first screening test in 2006, before the vaccine was marketed. The researchers discovered a 40 percent reduced risk of severe dysplasia in the 1993 birth cohort compared to the 1983 birth cohort.
"This means that fewer women have to be referred to a gynaecologist for further examination and have a tissue sample taken. Eventually, we also expect fewer to fall ill," says Lise Thamsborg, first author of the study.
The girls from the 1993 birth cohort were 15 years old when they received the vaccine. And the researchers expect the effect to be even more pronounced today, when girls are offered the vaccine at the age of 12.
"We expect the effect to be greater among those vaccinated at the age of 12, because very few have been sexually active at this age," says Lise Thamsborg.
However, the study did not only find a reduction in severe dysplasia. Contrary to expectation and the results of randomised trials, the women born in 1993 showed a higher level of mild dysplasia than the women born in 1983. The cause may be new technology introduced in 2006 for examining the cell samples that reveal cases of dysplasia.
"The new technology has led to fewer inadequate samples, and the samples are of a higher quality today. So the samples are more sensitive. This may be the cause," says Lise Thamsborg.
The next step for the researchers is to examine the tissue samples taken from women with dysplasia. The aim is to learn whether and how cases of mild and severe dysplasia have developed. If a woman suffers from severe dysplasia, a tissue sample is taken. It can reveal precursor lesions to cervical cancer. There are different levels of these precursor lesions. If a woman shows mild dysplasia, she is typically invited to an exam six months later to see whether the changes have stopped or developed further.
More information: Lise Holst Thamsborg et al, Impact of HPV-vaccination on outcome of cervical cytology screening in Denmark - a register based cohort study, International Journal of Cancer (2018). DOI: 10.1002/ijc.31568