Combining targeted radionuclide therapy and immunotherapy could improve melanoma survival
Research presented at the 2018 Annual Meeting of the Society of Nuclear Medicine and Molecular Imaging (SNMMI) shows that combining targeted radionuclide therapy with immunotherapy could improve the survival of patients with metastatic melanoma.
Immunotherapies, which use the patient's immune system to fight disease, have increased the survival of many advanced-stage cancer patients. However, for most patients their cancer will either not respond or initially respond but subsequently become resistant to immunotherapy.
"External beam radiotherapy has been shown to enhance cancer response when used in combination with immunotherapy in preclinical studies," explains Reinier Hernandez, Ph.D., of the University of Wisconsin-Madison. "Results can be significantly limited though in the presence of metastatic disease whereby distant tumors that are not targeted with external beam radiation may be relatively resistant to immune recognition and may even harbor suppressive immune cells that can limit the response of effector cells at the radiated tumor site."
He points out, "The true novelty of our work is that, this is the first time that targeted radionuclide therapy (TRT) has been shown to successfully synergize with immunotherapies. We have demonstrated in our animal model of melanoma that the combination of TRT and immunotherapy not only results in enhanced and durable treatment response but also a high cure rate and establishment of an anti-tumor immune memory."
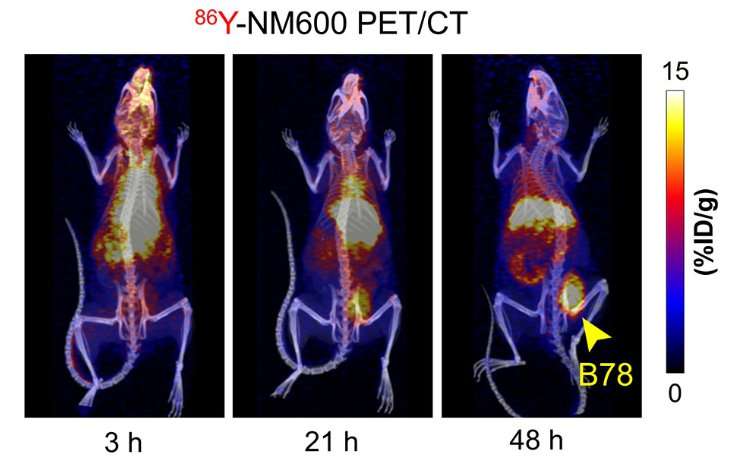
For the study, a mouse model of melanoma was used. The mice were initially administered yttrium-86 (86Y)-NM600 and had PET/CT scans at 3, 21 and 48 hours post injection to determine the proper activity of yttrium-90 (90Y)-NM600, the TRT agent, that would need to be injected to deliver a desired dose of radiation to the tumor microenvironment(s).
Hernandez explains, "Following intravenous injection of our TRT agent, it undergoes selective tumor uptake and prolonged retention, allowing for the precise delivery of the radiation dose to tumors wherever they are in the body—something that is unique to this form of radiation treatment. We have also demonstrated a low toxicity profile for normal organs and tissues at the low immunomodulatory radiation doses of NM600. Moreover, exposure concerns are minimized because NM600 can achieve this dose in the tumor microenvironment by delivering a beta-emitter like yttrium-90."
Mice treated with the combination of 90Y-NM600 and anti-CTLA-4 immunotherapy showed tumor regression and improved survival compared to other treatment groups, with 66 percent exhibiting a durable complete tumor response.
Hernandez and key coauthor Ravi Patel, MD, PHD, noted that this study took a multifaceted approach to combined TRT/immunotherapy research, which involved close collaboration with immunology, radiation oncology, dosimetry and imaging researchers at the University of Wisconsin-Madison. Such interdisciplinary collaboration is key to advancing new therapies, and NM600 has potential applications for diverse cancers.
Hernandez points out, "Unlike other receptor targeted TRT agents, whose efficacy is restricted to a limited number of patients, NM600 works through a mechanism that is ubiquitous to almost all cancers. This allows us to expand our combination approach to many other malignancies besides melanoma. We have already shown promising results in preclinical models of breast cancer and neuroblastoma and are currently testing this treatment paradigm in pancreatic and lung cancer models."
More information: Abstract 119: "Combination of Targeted Radionuclide Therapy and Checkpoint Blockade Augments Therapeutic Response in a Syngeneic Murine Model of Melanoma," Reinier Hernandez, Ph.D., Ravi Patel, Joseph J. Grudzinski, Ph.D., Peter Carlson, Raghava Sriramaneni, Ryan Brown, Bryan Bednarz, Paul Sondel, Zachary Morris, Jamey Weichert, Ph.D., University of Wisconsin-Madison, Madison, WI. SNMMI's 65th Annual Meeting, June 23-26, Philadelphia. jnm.snmjournals.org/content/59 … b0-bb6d-2bceae1a7d35