Why BACE inhibitors may be failing Alzheimer's trials
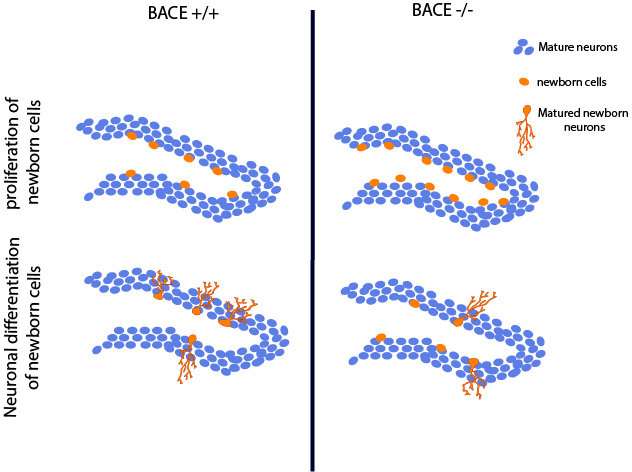
Completely blocking the activity of an enzyme that produces amyloid plaques observed in Alzheimer's disease (AD) interferes with the regulation of new neurons generated in the adult hippocampus, according to a study of mice published in eNeuro. These findings may help to explain why some recently developed AD drugs have failed in clinical trials.
Several recent trials of β-Site Amyloid Precursor Protein Cleaving Enzyme 1 (BACE1) inhibitors have been suspended due to their inability to improve AD symptoms despite reducing amyloid plaques or because of adverse side effects. Most researchers believe that BACE inhibitors fail on efficacy because they are stopping amyloid formation too late in the course of the disease while those failing as a result of safety issues are more likely related to drug structure. However, the recent Merck BACE inhibitor trial failure may have been due to safety issues owing to BACE inhibition itself. These BACE trial failures have not been attributed to a particular reason and may therefore stem, in part, from an incomplete understanding of BACE1's role in the healthy brain.
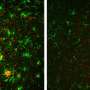
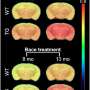
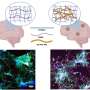
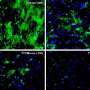
To address this knowledge gap, Rudolph Tanzi, Se Hoon Choi, and colleagues investigated whether BACE1 is involved in adult hippocampal neurogenesis—a process that may be responsible for the brain's ability to repair itself. Their study revealed that although mice which do not express BACE1 increase their production of neural stem cells, fewer of these cells reach maturity and could potentially compromise brain function in AD patients treated with a BACE inhibitor. The research suggests that partial, rather than complete, BACE inhibition may represent an alternative strategy for reducing AD pathology while maintaining adult neurogenesis.
More information: BACE1 regulates proliferation and neuronal differentiation of newborn cells in the adult hippocampus in mice, eNeuro, DOI: 10.1523/ENEURO.0067-18.2018