Startup developing novel treatment for life-threatening lung condition
A Purdue University-affiliated startup developing a novel treatment for a life-threatening lung condition known as acute respiratory distress syndrome, or ARDS, is taking part in a prestigious startup accelerator program where about $2 million in funding is available.
Spirrow Therapeutics (formerly Spiro Therapeutics) was selected to be a finalist from a pool of 1,600 applicants to take part in MassChallenge, the Boston-based startup accelerator that offers work space, an opportunity to connect with physicians and mentors and funding opportunities. The program will help as the researchers continue their work to create a novel nanopolymer surfactant that supplements lung inflation and blood oxygenation in ARDS patients.
"This is going to open a lot of opportunities for us to talk to experts," said Davis Arick, a co-founder of Spirrow Therapeutics who recently graduated from Purdue with a chemical engineering degree. "There are a lot of pulmonologists in the Boston area who work with ARDS regularly. We want to assemble an advisory board that can help us as we develop clinical studies."
Arick co-founded Spirrow Therapeutics with Kyle Kim, who earned a chemical engineering doctorate from Purdue, and You-Yeon Won, a Purdue chemical engineering professor. Originally interested in neonatal lung function, the researchers turned their attention to ARDS after discovering a greater need there.
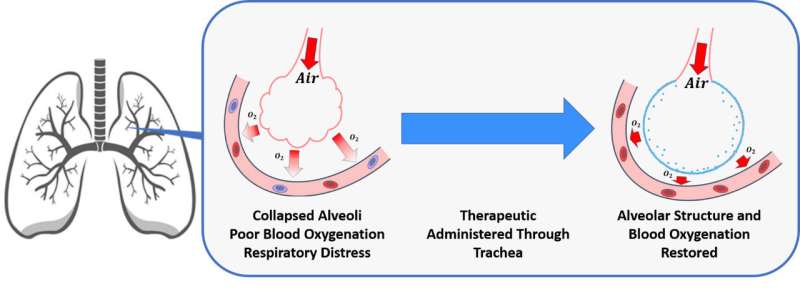
The United States has about 220,000 cases a year of ARDS, which deactivates the naturally occurring lung surfactant in patients. That causes a decreased ability to inflate the tiny air sacs in lungs called alveoli. When inadequately inflated, the alveoli cannot totally oxygenate a patient's blood stream, which can lead to organ failure and other health problems.
ARDS can be caused by pneumonia, septic shock, trauma, inhaling chemicals or other trauma. About 40 percent of people with ARDS die.
"So far, no ARDS treatments have improved mortality rates or long-term effects in ARDS patients," Arick said. "These patients don't have a great outlook for survival because there are not many treatments available. Our hope is to achieve this improvement by engineering a nanoparticle that forms a barrier in the alveoli and allows inflation, despite the deactivation mechanism's presence."
ARDS is currently treated through ventilation and a process known as extracorporeal membrane oxygenation, or ECMO.
"Over time, patients have less and less positive effects from these treatments because they force air into the lungs and overinflate them," Arick said.
Spirrow Therapeutics' treatment mimics the behavior of the natural surfactant in the lungs to work within the lungs' processes for inflation and oxygenation. The researchers also believe it is immune to the deactivation effect of ARDS. They say they are not trying to cure ARDS but are providing a way to help until the body begins producing the natural surfactant again and begins using it correctly.
Arick said the MassChallenge program, which began on June 25 and runs through Oct. 17, is somewhat similar to Purdue's Burton D. Morgan Business Model Competition, which Spirrow Therapeutics won in March to capture the $25,000 first prize. He said just being one of the finalists will help the company.
"The funds that are available at MassChallenge are great, but the connections are priceless," said Arick, a veteran of the entrepreneurial process.
Arick's introduction to the Purdue entrepreneurial ecosystem began at a young age. He won first place in a high school competition at the Purdue Foundry, which clinched his decision to attend Purdue. While at the university, he also took part in Purdue's Certificate in Entrepreneurship and Innovation Program.
"It was meaningful to have exposure to entrepreneurial programs while pursuing my engineering degree," he said. "Purdue is very progressive in how they teach business planning. Purdue is really good about this new movement to get out of the building and go talk to people to figure out who your customer is."
In the short term, Spirrow Therapeutics will focus on testing in preclinical trials, networking with physicians and advancing their business through MassChallenge. Following, the startup plans to begin the Food and Drug Administration approval process with the plan to eventually work with a pharmaceutical company to bring its nanopolymer surfactant to market.