August 31, 2018 report
Optogenetic profiling used to identify alterations in Ras signaling dynamics within cancer cells
A team of researchers at the University of California has found that altering the signals that cells use to communicate with one another can cause changes to transcriptional outcomes, possibly resulting in the development of tumors. In their paper published in the journal Science, the group describes using optogenetics to carry out extracellular signaling to learn more about its impact on cell proliferation. Walter Kolch and Christina Kiel with University Dublin offer a Perspective piece on the work done by the team in California in the same journal issue.
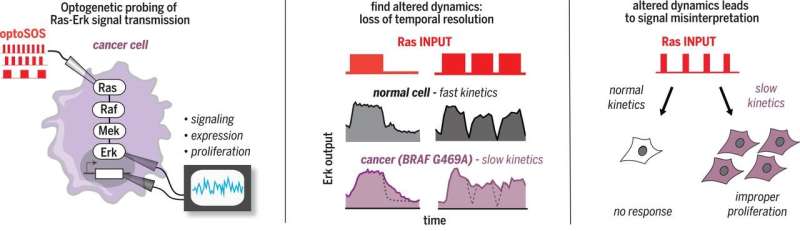
Kolch and Kiel note that signal transduction pathways (STPs) between cells serve to support the conversion of biochemical reactions into predictable outcomes—they even are able to do so in the presence of extraneous noise, which suggests they have some ability to discriminate between different signals. The pair further notes that such discrimination can be enhanced by introducing changes to signaling, such as altering rise time, duration, decay rate and amplitude. Past research efforts have shown, for example, that making such changes to STPs can cause rat pheochromocytoma cells to differentiate or proliferate. But, as they further note, it is still not clear how such signals are encoded and decoded. In this new effort, the team at UoC used a new approach to attempt to decipher STP codes.
The new approach involved using a light-controlled mechanism to activate and deactivate guanosine triphosphate (GTP) Ras on demand. GTP is a nucleotide that carries phosphates and pyrophosphates which are involved in directing chemical energy into specific biosynthetic pathways. More specifically, the team used an optogenetic tool that allowed them to turn on or off the expression of a kinase called RAS, or BRAF—a type of protein that is activated by growth factor receptors. In so doing, they found that they were able to affect transcriptional outcomes, which the researchers suggest, could lead to cell proliferation.
In addition to learning more about the coding used by STPs, the group also suggests that they have developed a new means for probing responses of signaling networks—a tool that could lead towards a better understanding of biological regulation.
More information: L. J. Bugaj et al. Cancer mutations and targeted drugs can disrupt dynamic signal encoding by the Ras-Erk pathway, Science (2018). DOI: 10.1126/science.aao3048
Abstract
The Ras-Erk (extracellular signal-regulated kinase) pathway encodes information in its dynamics; the duration and frequency of Erk activity can specify distinct cell fates. To enable dynamic encoding, temporal information must be accurately transmitted from the plasma membrane to the nucleus. We used optogenetic profiling to show that both oncogenic B-Raf mutations and B-Raf inhibitors can cause corruption of this transmission, so that short pulses of input Ras activity are distorted into abnormally long Erk outputs. These changes can reshape downstream transcription and cell fates, resulting in improper decisions to proliferate. These findings illustrate how altered dynamic signal transmission properties, and not just constitutively increased signaling, can contribute to cell proliferation and perhaps cancer, and how optogenetic profiling can dissect mechanisms of signaling dysfunction in disease.
© 2018 Medical Xpress