Farewell flat biology—Tackling infectious disease using 3-D tissue engineering
In a new invited review article, ASU Biodesign microbiologists and tissue engineers Cheryl Nickerson, Jennifer Barrila and colleagues discuss the development and application of three-dimensional (3-D) tissue culture models as they pertain to infectious disease. They describe these models as predictive pre-clinical platforms to study host-pathogen interactions, infectious disease mechanisms, and antimicrobial drug development.
The review, entitled "Modeling Host-Pathogen Interactions in the Context of the Microenvironment: 3-D Cell Culture Comes of Age" is the most comprehensive coverage of this topic to date. The publication explores in detail the history, development and application of 3-D tissue culture models of human intestine as predictive surrogate platforms to study host-microbe interactions that lead to infection and disease and charts a course for future innovation in drug and vaccine discovery.
A pioneer in the field, Nickerson was the first to apply 3-D tissue culture models to study infections caused by bacteria. The ultimate goal of researchers in this field is to develop fully functional, personalized, and predictive tissue model systems to understand the transition between health and infectious disease to bridge gaps in drug discovery. Indeed, the lack of physiologically relevant models of human tissues to study the disease process is believed to be a key contributing factor for the 90 percent failure rate of drugs in clinical trials.
As outlined in the review, the lack of representative and practical models of human tissues to study host-microbe interactions has led to a paradigm shift in the study of infectious disease in the laboratory toward the use of 3-D cell culture models that faithfully mimic key aspects of the structure and function of native tissues. Despite extensive research, many pathogens have no available vaccines or vaccines that are inadequate. These include leading causes of morbidity and mortality worldwide by pathogens responsible for diarrheal diseases, tuberculosis, AIDS, etc, many of which are multidrug resistant, and others which are potential tools of bioterrorists.
There is an urgent need to develop 3-D models of tissues that are structural and functional equivalents of their native tissues for use as infectious disease models. Insights gleaned from these models, which better replicate the interactions of hosts and pathogens in their natural microenvironments in the body, offer valuable insights into the mechanisms of infection and could aid in the design of more effective vaccines and therapeutic agents targeting a broad range of diseases.
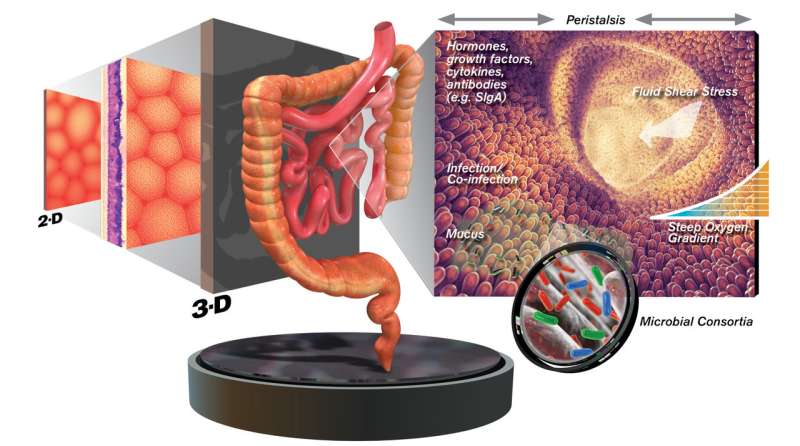
Pathogens and the cells and tissues they infect interact in a complex, three-dimensional world. Scientists hope to better understand these interactions in order to combat disease, yet the study of pathogen-host interactions has conventionally been carried out by examining the interplay of bacteria, viruses and other invaders with host cells grown on flat two-dimensional (2-D) surfaces as monolayers. While such studies have provided a wealth of information, there is a growing appreciation that many pathogen-host events are not accurately duplicated in 2-D models.
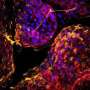
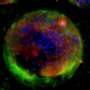
Nickerson's multidisciplinary team strives to reproduce key cellular, biochemical and physical tissue microenvironments found in the body to enable predictive infectious disease modeling, including 3-D tissue structure, multicellular complexity (incorporating immune cells), mechanical forces, pathogens and commensals (microbiome). Her team's establishment and characterization of a variety of 3-D tissue culture models and their application as predictive platforms for infectious disease research have provided specific examples of how the study of microbial pathogenesis can be advanced by using appropriate, biologically meaningful 3-D models. Their studies have shown that 3-D models respond to challenge with pathogens, their toxins, and antimicrobials in key ways that more accurately reflect the process in vivo as compared to flat 2-D monolayers.
Nickerson, a professor in the Biodesign Center for Immunotherapy, Vaccines and Virotherapy (BCIVV) and ASU's School of Life Sciences, and Barrila, lead author and an Assistant Research Professor in BCIVV, were joined by their Biodesign colleagues, Jiseon Yang, Karla Franco, Seth Nydam, Rebecca Forsyth, Richard Davis and Sandhya Gangaraju.
The study also relied on an international team of researchers, including Aurélie Crabbé of Ghent University, Belgium; C. Mark Ott from NASA Johnson Space Center; Carolyn Coyne from the University of Pittsburgh; and Mina Bissell from Lawrence Berkeley National Laboratory.
The invited review recently appeared in the ASM journal Infection and Immunity.
"By mimicking the structure and function of native tissues in the body, 3-D tissue models are powerful tools that serve as next generation platforms to unveil novel underlying principles of infectious disease, streamline vaccine/therapeutic discovery, predict how humans will respond to vaccines and other therapeutics, and increase likelihood of success in the most expensive part of vaccine development—human clinical trials," Nickerson says. "Moreover, the ability to create these models from patient samples offers exciting potential for personalized medicine."
According to Barrila: "Over the past several years we've seen an explosion of papers from scientists in the infectious disease community who are now using a variety of 3-D models to make important new discoveries to benefit human health. Our goal in writing this review was to highlight this exciting research and to discuss key factors to consider when modeling human tissue. We also included a brief historical perspective to help newer scientists in the field become aware of the rich history of 3-D modeling in other fields like cancer biology, so that they can leverage these past discoveries to inform their ongoing research."
Many types of pathogens favor the mucosal surface lining the intestinal tract as staging points for their infectious assault. However, this tissue has evolved complex mechanisms designed to thwart such attacks. These include a complex 3-D architecture, many different cell types (each geared to tissue-specific functions), physical forces (fluid shear and stretch/peristalsis), a vast array of antimicrobial compounds, oxygen gradients, and the largest microbiome in the body.
In a ceaseless arms race, pathogens have adapted to specific host structures and sought to exploit weaknesses in host defense mechanisms. Such invaders are able to monitor conditions of pH, temperature, oxygen, nutrients, hormones, inflammation, and physical forces to determine the most opportune time and place to activate infection processes and virulence.
Recreating the relevant details of these events in the laboratory has been a major challenge in the area of tissue engineering, particularly the spatial and temporal properties of tissue microenvironments, now known to be critical to the infection regime. Since tissues function in a three-dimensional context in our bodies, studying the infection process solely through the traditional use of cells grown as flat 2-D monolayers loses much of this vital information and can alter the behavior of both host cells and microbes under consideration.
This review provides an in-depth examination of the three most commonly used technologies for the development of 3-D models of human intestine, a major portal of entry for infectious pathogens and an important niche for commensal microbiota (microbiome). These technologies are the Rotating Wall Vessel (RWV) bioreactor, hydrogel-embedded models (organoids), and organ-on-a-chip. Collectively, these model systems are providing a more physiologically relevant and predictive framework for investigating infectious disease mechanisms and antimicrobial therapies at the intersection of the host, microbe and their local microenvironments. The review highlights seminal studies that have used these models to interrogate host-pathogen interactions and infectious disease mechanisms and presents this literature in the appropriate historical context.
As no model is perfect, the review also highlights the inherent advantages and limitations of these advanced model systems. For example, while all three technologies enable cells to undergo self-assembly into 3-D tissue-like structures and spontaneously differentiate into multiple epithelial cell types normally found in the intestine, only the RWV bioreactor and organ-on-a-chip currently incorporate physical forces (e.g., fluid shear and/or stretching, respectively) into their cell culture technologies.
The review also stresses that host cells and their invaders are only a small part of a complex ensemble of actors. Many cell types besides epithelial cells (e.g., immune cells, nerve cells, muscle cells and vasculature) have vital roles to play in the infection process, along with a complex microenvironment surrounding these cells. Physical/mechanical forces exerted on both host and pathogenic cells are also an integral part of a rich web of cellular communication mediating infection and defense.
The body's richly diverse complement of supportive or commensal organisms, collectively known as the microbiome, is also a critical contributor in mediating host-pathogen responses. These include a variety of bacteria, viruses, archaea, and eukaryotic cells. More than a thousand distinct species remain busily at work throughout the body, providing many forms of essential support, including the safeguarding of host cells from colonization by pathogens.
While recreation of the full extent of 3-D architecture, multicellular and biochemical complexity and array of mechanical forces normally found in the parental tissue has not yet been attained with any model system, ongoing studies are further enhancing these features, plus incorporating patient-specific immune cells and fecal microbiome, and achieving vascularization and innervation. These are the next steps in model advancement to better recapitulate the complex tissue microenvironment naturally encountered in the body by microbial pathogens.
The review further stresses that 3-D modeling of host-pathogen interactions is still in its formative stages, with many questions unanswered. Continued multidisciplinary development of such models thus offers hope for a more profound understanding of the underlying dynamics of infection and disease that will drive basic research discoveries and improved treatments and preventive strategies.
More information: Jennifer Barrila et al, Modeling Host-Pathogen Interactions in the Context of the Microenvironment: 3-D Cell Culture Comes of Age, Infection and Immunity (2018). DOI: 10.1128/IAI.00282-18