Rethinking Alzheimer's disease therapeutic targets using gene-based tests

Alzheimer's disease (AD) is a devastating condition with no known cure. Existing drugs only alleviate symptoms. Given repeated and costly drug failures, CUNY SPH Professor Mary Schooling and colleagues assessed systematically whether approved and investigational AD drugs are targeting products of genes strongly associated with AD and whether these genes are targeted by existing drugs for other indications which could be re-purposed.
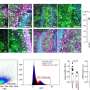
Schooling and her team identified genes strongly associated with late-onset AD from the loci of genetic variants associated with AD at genome-wide-significance and from a gene-based test applied to the most extensively genotyped late-onset AD case (n = 17,008)-control (n = 37,154) study, the International Genomics of Alzheimer's Project. They used three gene-to-drug cross-references, Kyoto Encyclopedia of Genes and Genomes, Drugbank and Drug Repurposing Hub, to identify genetically validated targets of AD drugs and any existing drugs or nutraceuticals targeting products of the genes strongly associated with late-onset AD.
A total of 67 autosomal genes were identified as strongly associated with late-onset AD. Existing approved or investigational AD drugs did not target products of any of these 67 genes. Drugs for other indications targeted 11 of these genes, including immunosuppressive disease-modifying anti-rheumatic drugs targeting PTK2B gene products.
The researchers found that approved and investigational AD drugs are not targeting products of genes strongly associated with late-onset AD. However, other drugs targeting products of these genes exist and could perhaps be re-purposing to combat late-onset AD after further scrutiny.
"This study shows the value of systematically searching bioinformatics resources to discover new leads concerning the prevention or treatment of complex diseases," Schooling says.
More information: Man Ki Kwok et al. Re-thinking Alzheimer's disease therapeutic targets using gene-based tests, EBioMedicine (2018). DOI: 10.1016/j.ebiom.2018.10.001