Bipolar structure for nerve cell migration
The cerebral cortex is responsible for a large number of complex brain functions, ranging from the perception of sensory stimuli through alertness, memory and language to consciousness. Neurobiologists at the Max Planck Institute for Experimental Medicine in Göttingen have decoded a new regulatory principle that controls the development of the complex neuronal architecture in the cerebral cortex.
The human brain, like that of all other mammals, is dominated by an outer layer rich in nerve cells known as the cerebral cortex. It consists of six layers of nerve cells that are linked with each other and other parts of the brain in a highly complex manner and control all complex brain functions.
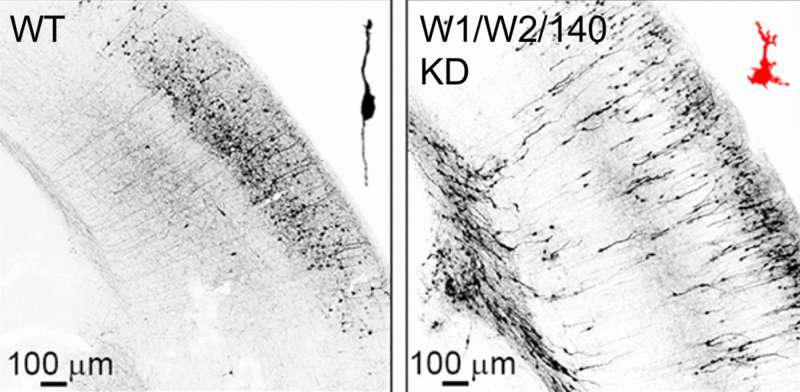
The formation of the elaborately layered nerve cell architecture in the cerebral cortex begins at a relatively early stage of embryonic development and is not completed until quite long after birth. First, new nerve cells form on the underside of the developing cerebral cortex, where they take on a streamlined – or bipolar – structure, migrate outwards perpendicular to the surface and finally stop in their target region, where they take on the form typical of nerve cells and develop numerous processes, enabling them to connect with other nerve cells.
The formation of the bipolar structure by the developing nerve cells, their so-called polarization, and their subsequent migration to the developing cerebral cortex are controlled by numerous intracellular regulatory processes that are in turn the target of a whole series of extracellular signal processes. Errors during these development processes lead to malformations of the cerebral cortex and almost always have serious consequences for affected patients, one example being lissencephaly.
Interleaved genes
Together with Victor Tarabykin from the Charité in Berlin and collaboration partners in Japan, Mateusz Ambrozkiewicz and Hiroshi Kawabe from the Max Planck Institute for Experimental Medicine in Göttingen have now discovered a new regulatory principle that controls the polarization and migration of new nerve cells. With the help of genetically manipulated mice, the discovered that the coordinated activity of the so-called ubiquitin ligases WWP1 and WWP2, which trigger the degradation of proteins, and the so-called micro RNA miR140, which inhibits the production of new proteins, plays a key role in the polarization and migration of nerve cells while the cerebral cortex is developing. What is particularly interesting is that the genes involved are in part interleaved, are controlled in a similar way, and jointly regulate related processes in developing nerve cells.
"Besides having discovered a new regulatory process in the development of the brain, we are particularly fascinated by the fact that the miR140 gene is embedded in the WWP2 gene," says study leader Hiroshi Kawabe. This interleaving of protein coding genes and micro RNA genes is rare but not unusual, and it has long been suspected that such gene structures indicate that the respective gene products have a common function. "However, we have now succeeded in proving that this is actually the case. The production of WWP2 and miR140 is coordinated by the nerve cells and both jointly support the cellular polarization process."
More information: Mateusz C. Ambrozkiewicz et al. Polarity Acquisition in Cortical Neurons Is Driven by Synergistic Action of Sox9-Regulated Wwp1 and Wwp2 E3 Ubiquitin Ligases and Intronic miR-140, Neuron (2018). DOI: 10.1016/j.neuron.2018.10.008