A look at how colds and chronic disease affect DNA expression

We're all born with a DNA sequence that encodes (in the form of genes) the very traits that make us, us—eye color, height, and even personality. We think of those genes as unchanging, but in reality, the way they are expressed, or turned into proteins, which are the workhorses of our molecular selves, is regularly in flux.
Many of those fluctuations remain little understood, including how a disease like a cold or a more complex chronic condition affects gene expression.
But Michael Snyder, Ph.D., professor and chair of genetics at Stanford, has been working to change that. He has been tracking how his body is changing on a molecular level in response to diseases, including diabetes, which Snyder has.
Now, after consistently sampling his own DNA and RNA (a molecular cousin of DNA that provides insights into gene activity), Snyder has spotted some interesting patterns that track with illness.
While the research only accounts for one person, it provides an unprecedented window into the molecular machinery that churns inside the body when it has a cold or grapples with a chronic disease. The study was published in Nature Medicine.
Over the course of three years, Snyder came down with a cold or flu six times, and had two notable increases in blood sugar that rose to diabetic levels. During every bout of viral illness, Snyder saw dramatic changes in his "transcriptome," a catalog of RNA molecules that indicates levels of gene activity.
During each illness, a core set of genes known to contribute to the immune response were revved up, which makes sense, but each viral infection also triggered activation of its own separate group of genes too. That could reflect the fact that the infections were caused by different virus strains.
After Snyder recovered from each bug, his transcriptome settled back into its normal groove, with immune-response genes tamping down activity. All of this, he said, was expected.
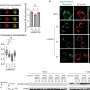
The surprise came when Snyder studied epigenetic changes across his genome, specifically, chemical modifications called methylation. (The prefix "epi" means on or upon, and epigenetics refers to a collection of molecules that latch onto DNA—rather than changing the sequence itself—to change the way it's expressed.)
DNA analysis showed that his methylation patterns generally only changed twice during the three years—both times just before Snyder had an upswing in blood sugar. In other words, his DNA was modified during the onset of his glucose dysregulation and diabetes spikes. Many of these changes were near genes known to function in metabolic control.
"Methylation is well known to be associated with a change in gene activity—the presence of methyl groups usually shuts off gene expression," Snyder said. "We're thinking that the regulatory sequence of these metabolic genes are being perturbed, and that's causing dysregulation in my glucose, contributing to my diabetes."
What's intriguing, he continued, is that the methylome starts changing prior to the glucose level spikes. "So because it's occurring a little bit before, it's a sign that the methyl changes might actually be somewhat responsible for, or contribute to the dysregulation of glucose." He noted that once his glucose levels dropped, the methylation did too.
"For the first time, we see that widespread changes occur during onset of diabetes," said Dan Xie, Ph.D., a former postdoctoral fellow at Stanford. The information, Snyder adds, could be important to understanding the different types of diabetes, why it's difficult to reverse, and could even play into how the disease is treated in the future.
Snyder's been monitoring himself in this way for eight years, although the study only reported on three. During that time, Snyder has taken periodic samples of his DNA and RNA through blood draws. They weren't on a strict schedule, but he made sure to increase collection frequency when he felt like he was coming down with a bug, since the goal was to see, in as much detail as possible, how his body's molecular landscape shifted in response to a pathogen invader or uptick in diabetes.
The next step is to see how these findings hold up when compared to other individuals. And while it's not plausible to monitor a large group of people as closely as Snyder monitored himself for as long as he did, he says that he's gearing up to analyze data from 100 people who've been participating in a similar study for a number of years.
More information: Rui Chen et al. Longitudinal personal DNA methylome dynamics in a human with a chronic condition, Nature Medicine (2018). DOI: 10.1038/s41591-018-0237-x