This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New mRNA cancer vaccine triggers fierce immune response to fight malignant brain tumor
In a first-ever human clinical trial of four adult patients, an mRNA cancer vaccine developed at the University of Florida quickly reprogrammed the immune system to attack glioblastoma, the most aggressive and lethal brain tumor.
The results mirror those in 10 pet dog patients suffering from naturally occurring brain tumors whose owners approved of their participation, as they had no other treatment options, as well as results from preclinical mouse models. The breakthrough now will be tested in a Phase I pediatric clinical trial for brain cancer.
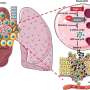
Reported May 1 in the journal Cell, the discovery represents a potential new way to recruit the immune system to fight notoriously treatment-resistant cancers using an iteration of mRNA technology and lipid nanoparticles, similar to COVID-19 vaccines, but with two key differences: use of a patient's own tumor cells to create a personalized vaccine, and a newly engineered complex delivery mechanism within the vaccine.
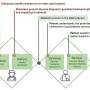
"Instead of us injecting single particles, we're injecting clusters of particles that are wrapping around each other like onions, like a bag full of onions," said senior author Elias Sayour, M.D., Ph.D., a UF Health pediatric oncologist who pioneered the new vaccine, which like other immunotherapies attempts to "educate" the immune system that a tumor is foreign.
"And the reason we've done that in the context of cancer is these clusters alert the immune system in a much more profound way than single particles would."
Among the most impressive findings was how quickly the new method, delivered intravenously, spurred a vigorous immune-system response to reject the tumor, said Sayour, principal investigator of the RNA Engineering Laboratory within UF's Preston A. Wells Jr. Center for Brain Tumor Therapy and a UF Health Cancer Center and McKnight Brain Institute investigator who led the multi-institution research team.
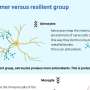
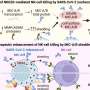
"In less than 48 hours, we could see these tumors shifting from what we refer to as 'cold'—immune cold, very few immune cells, very silenced immune response—to 'hot,' very active immune response," he said.
"That was very surprising given how quick this happened, and what that told us is we were able to activate the early part of the immune system very rapidly against these cancers, and that's critical to unlock the later effects of the immune response."
Glioblastoma is among the most devastating diagnoses, with median survival around 15 months. The current standard of care involves surgery, radiation and some combination of chemotherapy.
The new publication is the culmination of promising translational results over seven years of studies, starting in preclinical mouse models and then in a clinical trial of 10 pet dogs that had spontaneously developed terminal brain cancer and had no other treatment options.
That trial was conducted with owners' consent in collaboration with the UF College of Veterinary Medicine. Dogs offer a naturally occurring model for malignant glioma because they are the only other species that develops spontaneous brain tumors with some frequency, said Sheila Carrera-Justiz, D.V.M., a veterinary neurologist at the UF College of Veterinary Medicine who is partnering with Sayour on the clinical trials. Gliomas in dogs are universally terminal, she said.
After treating pet dogs that had spontaneously developed brain cancer with personalized mRNA vaccines, Sayour's team advanced the research to a small Food and Drug Administration-approved clinical trial designed to ensure safety and test feasibility before expanding to a larger trial.
In a cohort of four patients, genetic material called RNA was extracted from each patient's own surgically removed tumor, and then messenger RNA, or mRNA—the blueprint of what is inside every cell, including tumor cells—was amplified and wrapped in the newly designed high-tech packaging of biocompatible lipid nanoparticles, to make tumor cells "look" like a dangerous virus when reinjected into the bloodstream and prompt an immune-system response. The vaccine was personalized to each patient with a goal of getting the most out of their unique immune system.
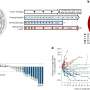
"The demonstration that making an mRNA cancer vaccine in this fashion generates similar and strong responses across mice, pet dogs that have developed cancer spontaneously and human patients with brain cancer is a really important finding, because oftentimes we don't know how well the preclinical studies in animals are going to translate into similar responses in patients," said Duane Mitchell, M.D., Ph.D., director of the UF Clinical and Translational Science Institute and the UF Brain Tumor Immunotherapy Program and a co-author of the paper.
"And while mRNA vaccines and therapeutics are certainly a hot topic since the COVID pandemic, this is a novel and unique way of delivering the mRNA to generate these really significant and rapid immune responses that we're seeing across animals and humans."
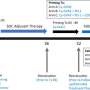
While too early in the trial to assess the clinical effects of the vaccine, the patients either lived disease-free longer than expected or survived longer than expected.
The 10 pet dogs lived a median of 139 days, compared with a median survival of 30 to 60 days typical for dogs with the condition.
The next step, through support from the Food and Drug Administration and the CureSearch for Children's Cancer foundation, will be an expanded Phase I clinical trial to include up to 24 adult and pediatric patients to validate the findings.
Once an optimal and safe dose is confirmed, an estimated 25 children would participate in Phase II, said Sayour, an associate professor in the Lillian S. Wells Department of Neurosurgery and the department of pediatrics in the UF College of Medicine, part of UF Health.
For the new clinical trial, Sayour's lab will partner with a multi-institution consortium, the Pediatric Neuro-Oncology Consortium, to send the immunotherapy treatment to children's hospitals across the country. They will do this by receiving an individual patient's tumor, manufacturing the personalized vaccine at UF and sending it back to the patient's medical team, said Sayour, co-leader of the Immuno-Oncology and Microbiome research program at the UF Health Cancer Center.
Despite the promising results, the authors said one limitation is continued uncertainty about how best to harness the immune system while minimizing the potential for adverse side effects.
"I am hopeful that this could be a new paradigm for how we treat patients, a new platform technology for how we can modulate the immune system," Sayour said.
"I am hopeful for how this could now synergize with other immunotherapies and perhaps unlock those immunotherapies. We showed in this paper that you actually can have synergy with other types of immunotherapies, so maybe now we can have a combination approach of immunotherapy."
Sayour and Mitchell hold patents related to the vaccine which are under option to license by iOncologi Inc., a biotech company born as a "spin out" from UF in which Mitchell holds interest.
More information: RNA aggregates harness the danger response for potent cancer immunotherapy, Cell (2024). DOI: 10.1016/j.cell.2024.04.003. www.cell.com/cell/fulltext/S0092-8674(24)00398-2