New generation of therapeutics based on understanding of aging biology show promise for Alzheimer's disease
A scientific strategy that explores therapeutic targets based on the biology of aging is gaining ground as an effective approach to prevent and treat Alzheimer's disease, according to research published in the December 7, 2018 online issue of Neurology.
A comprehensive review of the clinical trial landscape, including current agents being studied for the prevention and treatment of Alzheimer's disease (and other dementias), points to the need to develop and test drugs based on an understanding of the multiple effects of aging on the brain.
"Alzheimer's is a complex disease with many different factors that contribute to its onset and progression," says Dr. Howard Fillit, founding executive director and chief science officer of the Alzheimer's Drug Discovery Foundation (ADDF), senior author of the review paper. "Decades of research have revealed common processes that are relevant to understanding why the aging brain is vulnerable to Alzheimer's disease. New therapeutics for Alzheimer's disease will come from this understanding of the effects of aging on the brain."
The only approved medications for Alzheimer's disease relieve some symptoms but do not halt disease progression. New therapies that prevent, slow, or stop the disease are urgently needed to fight the growing Alzheimer's disease burden in the United States and around the world. And, aging biology provides numerous novel targets for new drug development for Alzheimer's disease, notes Dr. Fillit.
"Our success in fighting Alzheimer's disease will likely come from combination therapy—finding drugs that have positive effects on the malfunctions that happen as people age," says Dr. Fillit. "Combination therapies are the standard of care for other major diseases of aging, such as heart disease, cancer, and hypertension, and will likely be necessary in treating Alzheimer's disease and other dementias."
Increasing age is the leading risk factor for Alzheimer's disease, a progressive neurodegenerative disease that affects 5 million people in the United States and about 50 million globally. With a growing aging population, the Centers for Disease Control and Prevention projects the burden of Alzheimer's disease will nearly triple to 14 million people by 2060.
With aging, many biological processes go awry that have also been implicated in Alzheimer's disease. For example, as people age, they are more likely to have chronic systemic inflammation and neuroinflammation, which is associated with poorer cognitive function. Other aging malfunctions include impaired clearance of toxic misfolded proteins, mitochondrial and metabolic dysfunctions (associated with diabetes), vascular problems, epigenetic changes (changes in gene regulation without alterations in the DNA sequence), and loss of synapses (points of communication between neurons).
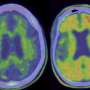
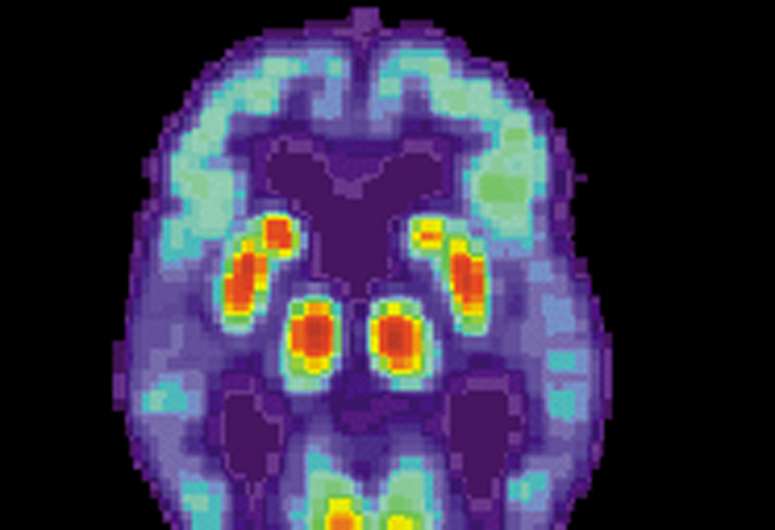
Later-phase (phase 3) trials are dominated by drugs targeting beta-amyloid and tau, the classic pathological hallmarks of Alzheimer's disease (of phase 3 trials, 52% are targeting amyloid or tau), but other strategies are gaining ground and are in phase 1 or 2 trials, according to the review paper.
Although therapeutic attempts to remove or decrease the production of beta-amyloid have been largely unsuccessful in altering the disease course of Alzheimer's disease, says Dr. Fillit, researchers learned important information from those clinical trials even if they didn't immediately result in treatments for Alzheimer's patients. And recent clinical trials suggest that problems with clearance of beta-amyloid may yet prove fruitful.
"It is currently not known if these classic pathologies (amyloid and tau) represent valid drug targets and if these targets alone are sufficient to treat Alzheimer's disease," says Dr. Fillit. "Targeting the common biological processes of aging may be an effective approach to developing therapies to prevent or delay age-related diseases, such as Alzheimer's."
More information: Neurology (2018). n.neurology.org/content/early/ … WNL.0000000000006745 , DOI: 10.1212/WNL.0000000000006745