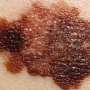
New study finds SkinVision detects 95% of skin cancer
An international group of researchers working with Erasmus University Medical Center (Erasmus MC) in the Netherlands determined that SkinVision—the first CE-marked skin cancer application based on extensive clinical trials—has a sensitivity of 95.1% in detecting the most common forms of skin cancer.
The research published in the Journal of the European Academy of Dermatology and Venereology (JEADV) evaluated the accuracy of the application for triaging skin lesions. The researchers found that the SkinVision algorithm has high sensitivity to detect skin cancer and can be a valuable tool for early skin cancer detection.
Early Recognition of Skin Cancer
Skin cancer is a growing problem, with currently between 2 and 3 million non-melanoma skin cancers and 132,000 melanoma skin cancers occurring each year globally. In fact, one in three cancers diagnosed is skin cancer. The good news is that 95% of skin cancers can be successfully treated if caught at an early stage.
As healthcare systems are adopting more innovative technology, there is increasing evidence that mobile health apps can help to save lives. SkinVision empowers individuals to take their skin health into their own hands by transforming their smartphone into a medical device for skin cancer risk assessment. So far, SkinVision has assisted in the finding of more than 35,000 skin cancer cases globally and managed to direct those people to a healthcare professional for further examination.
The service has the potential to triage those in need of treatment and could help to decrease both the average cost of treatment and the number of unnecessary doctor visits. The company has built up a customer portfolio of 1,200,000 users globally and a database of 3.5 million pictures of suspicious and benign skin spots.
"The field of mHealth apps for the risk assessment of skin cancer is undergoing rapid evolution. It is clear from this and other recent studies based on clinical images that the application of machine learning in large image databases leads to a high sensitivity to distinguish between malignant and benign skin cancer cases," said Tiago M. C. D. Marques, postdoctoral researcher at Erasmus MC, and one of the study authors.
To evaluate the sensitivity of the algorithm, researchers used 285 histopathologically validated skin cancer cases from two previously published clinical studies and the smartphone application user database. The specificity was computed on a separate set from the user base, which contained 6,000 clinically-validated cases.
"Providing the layperson with a highly sensitive, robust and easy-to-use method to check skin lesions for cancer has been the SkinVision research team's goal for the past seven years," said Andreea Udrea, a Ph.D. researcher at SkinVision, associate professor at University Politehnica of Bucharest and the lead author of the study. "I'm confident to say that SkinVision users benefit from the latest technological advancements that have been incorporated in a rigorous and clinically tested mHealth app."
In terms of specificity, Erasmus MC is currently preparing a prospective study for further clinical validation, which will be performed at the Outpatient Dermatology Department during the coming months.
More information: A. Udrea et al. Accuracy of a smartphone application for triage of skin lesions based on machine learning algorithms, Journal of the European Academy of Dermatology and Venereology (2019). DOI: 10.1111/jdv.15935