Researchers identify new therapeutic target for pulmonary fibrosis
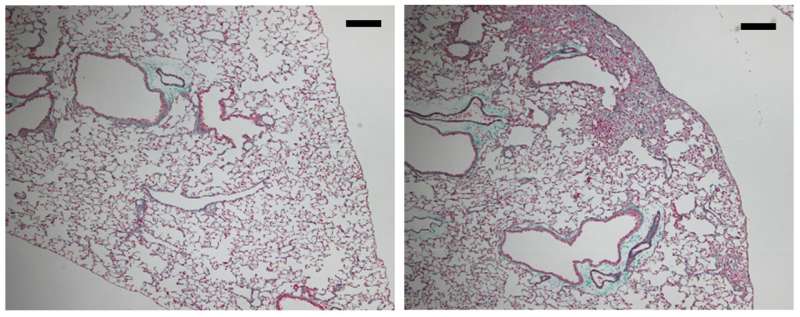
Researchers in Japan have identified a genetic mutation that causes a severe lung disease called idiopathic pulmonary fibrosis (IPF) by killing the cells lining the lung's airways. The study, which will be published October 10 in the Journal of Experimental Medicine (JEM), suggests that protecting these cells by inhibiting a cell death pathway called necroptosis could be a new therapeutic approach to treating IPF.
IPF is a progressive, and often fatal, lung disease characterized by a gradual buildup of scar tissue within the lungs, causing patients severe breathing difficulties. "The environmental and genetic factors underlying IPF are largely unknown, and there are no effective approaches available to reverse development of the disease," explains Dr. Koji Yasutomo from Tokushima University Graduate School of Medicine in Japan.
Dr. Yasutomo and colleagues identified two Japanese brothers who succumbed to IPF in their early thirties. Sequencing of the brothers' DNA revealed that they carried a mutation—dubbed T622C—in both copies of a gene called SFTPA1. This gene encodes a protein, surfactant protein A1, that is secreted by the cells lining the lung's alveoli, the tiny air sacs that mediate the exchange of oxygen and carbon dioxide. This usually helps to prevent the alveoli from collapsing and protects them from bacterial infection. Mutations in the SFTPA1 gene have been found in other patients with IPF, but how these mutations might cause disease is unclear.
The researchers generated mice carrying the T622C mutation and found that they, too, developed IPF. Similar to IPF patients, the animals' condition was fatally exacerbated by influenza A infection. "This strongly suggests that the mutation we identified in human SFTPA1 was the cause of IPF in this family," Yasutomo says.
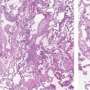
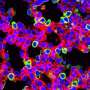
Yasutomo and colleagues determined that the T622C mutation in STFPA1 blocks the protein's release from alveolar cells, causing the cells to die via a process known as necroptosis. This form of cell death induces high levels of inflammation that is likely to increase the formation of scar tissue within the lung.
The researchers found that, instead of being secreted, the mutant form of SFTPA1 builds up inside the alveolar cells and activates a cellular stress pathway that boosts the levels of necroptosis-promoting proteins. Blocking this stress pathway, or otherwise reducing the levels of these necroptosis-promoting proteins, slowed the development of IPF in SFTPA1 mutant mice and allowed them to survive influenza A infection.
"Our study suggests that necroptosis is one of the crucial initiators of pulmonary fibrosis and that the necroptosis signaling pathway could be a potential target for its treatment," Yasutomo says. "The current focus for treating IPF is to block the activation of kinase enzymes within the fibrotic regions of the lung. In contrast, inhibiting necroptosis in alveolar cells would suppress earlier events in IPF progression, which would be more beneficial to patients."
More information: Takezaki et al., 2019. J. Exp. Med. jem.rupress.org/cgi/doi/10.1084/jem.20182351