Research: Hereditary mutation drives aggressive head and neck, lung cancers in Asian population
Researchers from the Cancer Science Institute of Singapore (CSI Singapore) at the National University of Singapore (NUS) have uncovered a genetic variant in a gene called MET that is responsible for more aggressive growth of head and neck, and lung cancers. A further probe into the finding revealed therapeutic strategies that could potentially target this genetic alteration, thereby paving the way for clinicians to develop better and more effective treatments for cancer patients of such profile.
The study, published in prestigious scientific journal Nature Communications on 25 March 2020, was conducted in close collaboration with clinicians from the National University Cancer Institute, as well as researchers from the National Cancer Centre Singapore and the Bioinformatics Institute at the Agency for Science, Technology and Research, Singapore.
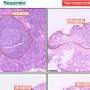
The MET gene encodes for a cancer-promoting protein that relays growth, survival and transmission of signals in cancer cells. In the study led by Professor Goh Boon Cher and Dr. Kong Li Ren from CSI Singapore, the team of researchers identified a form of MET protein, which showed ethnic preference with higher incidence among Asians, and is associated with poorer prognosis in patients diagnosed with head and neck squamous cell carcinoma or lung squamous cell carcinoma. Even though the MET variant does not seem to predispose an individual to cancer, it leads to more aggressive growth of cancers that have already developed.
Unlike other MET mutants, this genetic variant also does not appear to be inhibited by existing MET-blocking drugs that have been developed and approved in the clinical setting, prompting the researchers to conduct further investigation on the mechanism behind the genetic alteration.
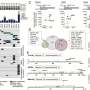
Leveraging the research team's multi-disciplinary expertise and state-of-the-art molecular modelling, the team found that the single amino-acid change in the MET receptor from the genetic alternation leads to preferential strong binding to another cancer promoting protein, HER2. Both proteins then work cooperatively to drive cancer aggression and enable cancer cells to survive therapies involving MET-blocking drugs.
"The mechanism of this MET variant is novel and unreported. This finding contributes to the growing evidence of the role of genetic variants in affecting clinical outcome, and underscores the importance of diving deep into our genetic inheritance in cancer research," said Dr. Kong, Research Fellow at CSI Singapore who initiated the study.
Knowledge of this unique mechanism also facilitated the team in identifying several HER2 inhibitors capable of blocking cancer progression caused by this genetic alteration using laboratory models.
Prof Goh, Deputy Director and Senior Principal Investigator at CSI Singapore, said, "Our study represents a conceptual advancement to cancer research, as we have shown that it is possible to block the activity of a cancer-driving gene by administrating a targeted therapy directed not against the mutant protein in question, but rather, a corresponding protein with which it binds to. The remarkable anti-tumour responses observed in our experimental models, coupled with the availability of FDA-approved HER2 inhibitors also presents a huge opportunity for clinicians to improve disease outcome of this genetic alteration via precision medicine."
More information: Li Ren Kong et al. A common MET polymorphism harnesses HER2 signaling to drive aggressive squamous cell carcinoma, Nature Communications (2020). DOI: 10.1038/s41467-020-15318-5