An optical brain-to-brain interface supports information exchange for locomotion control
Communications between two human or animal individuals conventionally depend on sensory systems for vision, audition, olfaction, or touch. Science fiction has popularized the potentials of directly transmitting information between brains for locomotor control. For example, in the 2009 film "Avatar," humans use their minds to remotely control the brains of Na'vi-human hybrids to navigate in the real world. Several recent studies proposed the possibility of retrieving electrophysiological signals from one brain to influence the neuronal activity in another brain through electrical or transcranial magnetic stimulation, suggesting the exciting concept of direct information exchange between brains through the Brain-to-Brain interfaces (BtBIs).
However, BtBIs have thus far required the use of demanding techniques for long-term, multi-channel recordings to decode the information from an encoder individual, and has been limited by low rates of information transmission to a target neural circuit. Multi-channel single-unit recordings are technically challenging and often lack cell-type specificity. EEG recordings are inaccessible to subcortical areas to precisely decode specific intention.
Moreover, EEG recordings of steady-state visually evoked potentials require external visual stimulation to generate the brain activity rather than the internal neural activity. Another challenge lies in the need of feeding the electrophysiological information, once decoded, into correct cell types and neural circuits in the target brain. Due to these technical limitations, the information transfer rates were often in the low range of 0.004-0.033 bits/s. Using a BtBI to control locomotion appears to be particularly difficult, since locomotion involves frequent starts, stops, and continuous changes in velocity at a sub-second scale.
Recently, Dr. Minmin Luo's lab published a research article entitled "An Optical Brain-to-brain Interface Supports Rapid Information Transmission for Precise Locomotion Control," in the journal Science China Life Sciences. In this work, the authors established an optical BtBI that supports rapid information transmission for precise locomotion control, thus providing a proof-of-principle demonstration of fast BtBI for real-time behavioral control.
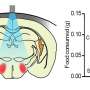
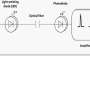
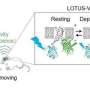
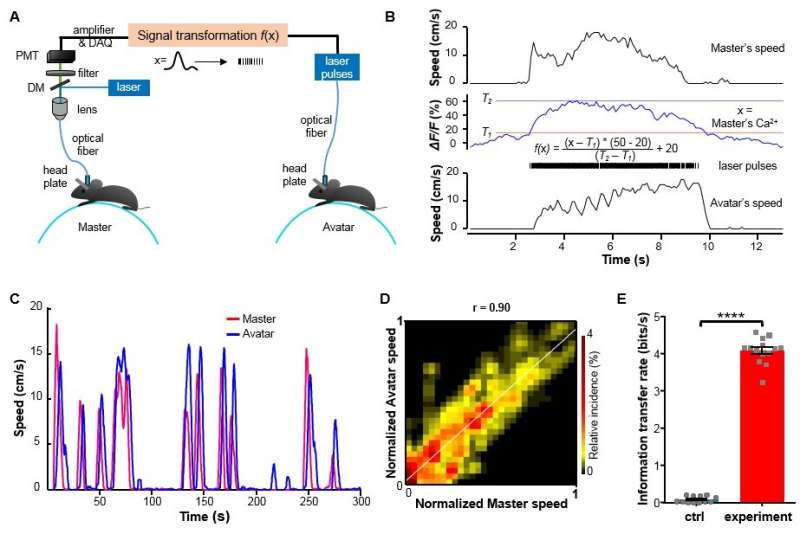
In this study, the authors demonstrated an optical BtBIs that used fiber photometry to record the population Ca2+ signals of NI neurons from the Master mouse, and then transformed the signals to blue laser pulses, and finally delivered the laser pulses into the NI of the Avatar mouse. This optical BtBI directed the Avatar mice to closely mimic the locomotion of their Masters with information transfer rate about three two orders of magnitude higher than previous BtBIs.
This study emphasizes the importance of choosing appropriate neural circuits and of choosing suitable circuit-probing technologies when building a high-performance BtBI. First, the choice of brain structures is important for implementing task-relevant BtBIs. Here the authors collected neuronal signals that precisely report locomotor state and control locomotor speed from the genetically-identified NMB neurons in the NI of the pons. Second, the choice of fiber photometry of Ca2+ signals offers several advantages: 1) it stably records the population neuronal activity of specific cell-type that performs similar functions; 2) it has high signal-to-noise ratio (SNR); 3) it is easy to implement, since it bypasses the challenging task of multi-channel single-unit recording from behaving animals and obviates the need for the extensive decoding of information from large datasets. Finally, the authors used optogenetic stimulation, which also enjoys the advantage of fine-tuning the activity of a genetically defined set of neurons in a given brain area.
In summary, this study demonstrates an optical brain-to-brain interface that supports rapid information transmission for precise locomotion control, and represented a major step toward realizing the full potential of BtBIs.
More information: Lihui Lu et al, An optical brain-to-brain interface supports rapid information transmission for precise locomotion control, Science China Life Sciences (2020). DOI: 10.1007/s11427-020-1675-x