Newly discovered interactions between proteins can reduce DNA damage and cancer development

Researchers at the Karolinska Institutet have discovered a new interaction between two proteins that reduce the risk of DNA damage and cellular cancer development. The findings on the proteins RTEL1 and Poldip3 are published in the journal Genes & Development.
Before a cell divides to form two new cells, it must copy its entire genetic information, so-called replication. A variety of proteins ensure that this is done correctly so that no changes are introduced to the DNA molecule as it could have consequence, such as cancer development or cell death. DNA: RNA hybrids—R loops—are a type of structure that can hinder replication. R loops have regulatory functions in the cell, but when they occur at the wrong times, they can lead to DNA damage.
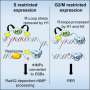
In the study, the researchers identified two proteins, RTEL1 (Regulator of telomere length 1) and Poldip3, which cooperate and prevent R loop formation. It is previously known that RTEL1 (Regulator of telomere length 1) is involved in maintaining telomeres, the ends of the chromosomes that are important for the cell's lifespan, and in the replication of the cell's DNA. Poldip3 is involved in both replication and RNA biology. The researchers showed that the proteins recruit each other to DNA after treatments with compounds that induce DNA damage.
Risk of DNA damage or cancer in cells without RTEL1 or Poldip3
In cells lacking RTEL1 or Poldip3, the prevalence of R-loops increases primarily in DNA regions with active replication. Even during replication, certain genes need to be transcribed and when these two processes occur simultaneously, the risk of R-loops is increased, which can hinder replication and cause genomic instability.
"We believe that RTEL1 and Poldip3 are especially important in preventing these R-loops. Two studies recently published in Nature Structural and Molecular Biology also support the hypothesis that RTEL1 prevents transcriptional replication conflicts and R-loops," says Andrea Björkman, the study's author and project leader in the Jiri Bartek group at the Department of Medical Biochemistry and Biophysics.
More information: Human RTEL1 associates with Poldip3 to facilitate responses to replication stress and R-loop resolution. Genes & Development, online June 2020