Engineered killer immune cells target tumours and their immunosuppressive allies

Scientists have engineered natural killer immune cells that not only kill head and neck tumor cells in mice but also reduce the immune-suppressing myeloid cells that allow tumors to evade the immune response, according to a new study in eLife.
The engineered cell therapy could be used as an alternative approach for treating cancer in patients for whom previous immunotherapy based on the activation of T cells has failed. These findings are reported by researchers at the U.S. National Institutes of Health (NIH) in Bethesda, Maryland.
In recent years, treatments called T-cell therapy or CAR-T cell therapy have been approved to treat blood cancers, and many others are now in development for other forms of cancer. However, these T-cell therapies rely on the ability to reprogram a patient's own T cells to express a chimeric antigen receptor (CAR) that targets tumor cells. This process of reprogramming a patient's own T cells is expensive and laborious.
High affinity natural killer cells (haNKs) represent potential 'off-the-shelf' cell therapies that do not rely on reprogramming a patient's own immune cells. The same cells could be produced in mass and potentially given to anyone. But the presence of immune-suppressing myeloid cells in the tumor microenvironment remains a barrier to effective immunotherapy, including haNK cell-based treatment.
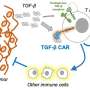
To address this barrier, researchers from the NIH's National Institute on Deafness and Other Communication Disorders (NIDCD) and National Cancer Institute have utilized haNKs expressing a CAR that targets a molecule called programmed death ligand 1 (PD-L1). PD-L1 is a well-known culprit that cancer and immunosuppressive myeloid cells produce in high amounts to dampen down the immune system.
Led by senior author Clint Allen, Principal Investigator, Section on Translational Tumor Immunology, NIDCD, the team tested the engineered PD-L1 haNKs versus ordinary haNKs against human and mouse head and neck cancer cells. They found that the haNKs expressing the PD-L1 CAR kill mouse and human tumor cells to a greater degree than haNKs without the CAR, and that this ability was retained even if they had already been exposed to cells carrying PD-L1 before. This is important because natural killer cells are known to become 'exhausted' after killing target cells.
In mice with head and neck tumors, the haNK cell-based therapy cured the mice in 30% of cases and slowed the growth of tumors in the rest of the mice, without causing toxicity. Treatment with haNKs also reduced the numbers of immunosuppressive myeloid cells that carry PD-L1 within the tumor, while having no effect on other immune-boosting white blood cells.
To investigate whether this effect on the immune cells also occurred in patients, the team incubated white blood cells from people with advanced head and neck cancer with the PD-L1 haNK cells. As they saw in the mice, the immunosuppressive myeloid cells that carry PD-L1 were significantly reduced after treatment with the PD-L1 haNK cells. This suggests that this treatment can both directly kill tumor cells and remove the immunosuppressive myeloid cells that prevent conventional immunotherapies from working.
These findings suggest that haNK cells expressing a PD-L1 CAR may overcome some of the limitations of conventional immunotherapy that relies on T-cell activation, and could be used in patients who are predicted to be insensitive to or have failed existing immunotherapy treatments. The researchers say the next steps would be to take this treatment into the clinic to explore the safety of PD-L1 haNKs in people with advanced or recurring cancer, and to see whether combining haNK cell therapy with other immunotherapies that activate T cells can enhance treatment response.
More information: Yvette Robbins et al, Tumor control via targeting PD-L1 with chimeric antigen receptor modified NK cells, eLife (2020). DOI: 10.7554/eLife.54854