Alzheimer's research strengthens evidence for reanalyzing data in clinical trials that 'failed'

New research on Alzheimer's disease led by the University at Buffalo reveals strong evidence that clinical trials on drugs that appeared not to benefit patients with the disease should now be reanalyzed in light of discoveries about a human-specific gene that divides the population into a one-to-three ratio.
The research builds on previous work by the UB group. That work found that this gene, CHRFAM7A, which is unique to humans but isn't present in everyone in its active form, can determine whether or not they will respond to three out of the four Alzheimer's disease treatments approved by the Food and Drug Administration that increase acetylcholine, a neurotransmitter involved in learning and memory.
That work illuminated why promising mice studies of potential Alzheimer's drugs targeting the alpha 7 nicotinic receptor appeared to be unsuccessful when they advanced to human trials.
"This uniquely human gene modifies the alpha 7 nicotinic receptor and as a consequence, drugs that are optimized in mice are insufficient for 75% of the population that has the active CHRFAM7A gene and the humanized receptor," said Kinga Szigeti, MD, Ph.D., corresponding author, director of UB's Alzheimer's Disease and Memory Disorders Center, professor of neurology in the Jacobs School of Medicine and Biomedical Sciences at UB and part of UBMD Neurology.
"This underlying, previously unknown genetic heterogeneity, especially with this ratio, undermined drug development efforts," she explained. "Our data indicate that drugs that target the nerve cells in which acetylcholine acts as a neurotransmitter, known as the cholinergic system, may benefit 25% of the individuals and now we can identify who they are. For Alzheimer's, if we can treat 25% of patients, that is 1.5 million people. That would be a major advance."
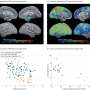
The research was conducted on induced pluripotent stem cells converted from the skin cells of patients with Alzheimer's disease and validated in clinical data from a 10-year, longitudinal, multicenter cohort study by the Texas Alzheimer Research and Care Consortium (TARCC) on 345 Alzheimer's patients, as well as data from the UB cohort.
The new research, published today in EBioMedicine, reports results from the iPSC model and the pharmacogenetic studies.
"This system that we created models the 25% of patients who could respond to the cholinergic drugs that supposedly failed in clinical trials, and contrasts them with the 75% whom likely will need a different approach," Szigeti said. "This is the best evidence so far that those clinical trials should be reanalyzed."
The research also provides evidence that individualizing treatment to patients' genotypes will be key to attacking Alzheimer's disease.
"This is the first proof of concept study that genotype and mechanism specific treatment is feasible in AD," the paper reports.
The researchers have concluded, instead of one disease for which a single drug is the solution, Alzheimer's disease, may, like cancer, represent a number of diseases, each of which will require its own targeted treatment.
"It takes a simple blood test to determine a patient's genotype, which will reveal whether or not a patient will respond to this class of drugs," Szigeti said.
The results of the UB study would need to be validated in larger, double-blind clinical trials, Szigeti said, noting that this can be achieved by reanalyzing the completed trials, saving millions of dollars.
More information: Kinga Szigeti et al. CHRFAM7A: A human specific fusion gene, accounts for the translational gap for cholinergic strategies in Alzheimer's disease, EBioMedicine (2020). DOI: 10.1016/j.ebiom.2020.102892