Epigenetics key to daily production of 10 billion blood cells without mistakes
Just one in 2,000 bone marrow cells are hematopoietic stem cells (HSC), but these are the source of the ten billion blood cells humans make every day. In a new study published today, researchers from the Centre for Genomic Regulation (CRG), in Barcelona, show that the epigenetic regulator Phf19 is essential for HSC differentiation, and in its absence blood tissue is imbalanced and equivalent to what naturally occurs with aging.
Blood is a complex tissue made up of different specialized cells (white blood cells, red blood cells, platelets and more types) that perform various essential functions ranging from the transport of oxygen and nutrients, maintaining body temperature or regulating the immune system. All blood cells are generated from HSCs, precursor cells that are stored in the bone marrow.
Blood diseases affect a large part of the worldwide population. One in four people suffer from anemia, 17 out of 100,000 have hemophilia and 2.5 out of 100 tumors that are detected worldwide are leukemic in nature. All these abnormalities are the result of malfunctioning blood cells or imbalances in the composition of blood tissue.
Maintaining blood tissue balance is complex because each cell has a different lifespan. White blood cells last five to 20 days while red blood cells last 120 days. To renew each of these cell types in the face of a threat such as a wound or bleeding, HSCs, which normally remain dormant, begin to divide and produce specialized cells to maintain the balance of blood tissue.
The process of transition from HSCs to specialized cells is regulated by epigenetic factors, that is, proteins that determine which genes that are expressed at each moment and cellular tissue. For this reason, the group of CRG researchers led by Luciano Di Croce studied the Phf19 gene, which is part of a large group of epigenetic regulators.
"Bone marrow cells are constantly dividing and are very sensitive to environmental factors such as radiation or chemotherapy," explains Arantxa Gutiérrez, co-first author of the article. "Any changes that affect the gene expression of HSCs, including epigenetic factors, can have a major impact on blood balance due to the plasticity of these cells."
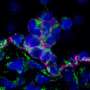
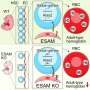
In the study published today in Science Advances, CRG researchers describe the role of the epigenetic regulator Phf19 in maintaining HSCs. They genetically modified mice to remove the Phf19 gene without affecting its normal lifespain. Without the gene the regions of the genome containing the genes responsible for HSC differentiation were more compacted, and did not express. As a consequence, HSCs remained quiescent and did not differentiate as much into specialized cells.
The researchers found that under normal conditions, the mouse's life was healthy, but in certain situations such as undergoing transplants or aging, the difficulty of producing differentiated cells compromised the proper functioning of blood tissue. In the long term, animals lacking Phf19 accumulated disorders in the composition of blood compatible with the early stages of leukemia.
"The vast majority of studies that have been done so far have removed all the biochemical functions from the multiprotein complex of which Pfh19 is part of. We have taken a subtler approach by eliminating a single gene, allowing us to discover a unexpected role in the regulation of MHC," explains Luciano Di Croce, lead author of the study.
"Until now it was known that aging conditioned the quantity and activity of the PHF19 protein and other proteins of the complex that it is part of, both in mice and in humans" says Di Croce. What was not known is that PHF19 controls HSC activation to begin to specialize.
According to Pedro Vizán, co-first author of the study and recipient of a grant from the Asosiación Española Contra el Cáncer (AECC) for this research, "the study of the processes by which stem cells regulate genetic information to produce specialized cells is essential for understanding how cells acquire pluripotence and proliferation capacity, key characteristics to form tumors." Now we have seen that "the lack of Phf19 increases the probability of suffering disorders in the composition of the blood. That is why we are studying its possible role as a precursor of tumors or whether it would be an anticancer therapeutic target."
More information: "The Polycomb-associated factor Phf19 controls hematopoietic stem cell state and differentiation" Science Advances (2020). advances.sciencemag.org/lookup … .1126/sciadv.abb2745