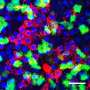
Scent-sensing cells have a better way to fight influenza
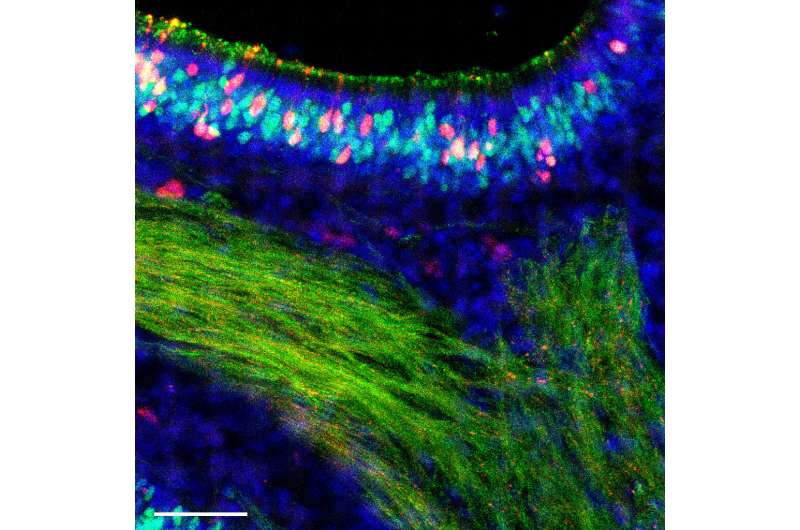
Influenza researchers have long focused most of their efforts on the epithelial cells lining the lungs because these are the cells that become infected and killed while producing new copies of the virus.
But other cells lining the upper airways are exposed to viruses in the same amounts and somehow aren't as likely to be killed by infection. Is it because of something the virus does, or something those cells do?
"So you're standing on the bus, somebody sneezes, and you breathe something in," said Nicholas Heaton, an assistant professor of molecular genetics and microbiology in the Duke School of Medicine. "But where does that virus go? It's certainly not somehow sneaking all the way down to the bottom of your lungs before it encounters a cell. It's hitting somewhere in your nose."
In a paper appearing Sept. 1 in Cell Reports, a collaboration between Heaton's team and the laboratory of Ashley Moseman in Duke immunology reports on the remarkably robust immune response of olfactory sensory neurons, the smell receptors that line the nose, where a virus might first be encountered. Their finding reveals not only a successful strategy against infection, it points out the diversity of immune responses from one kind of cell to another, Heaton said.
Of all the cells in the nose, olfactory neurons pose a unique danger because they form a direct connection from the airway to the nervous system. Any virus gaining control of that cell potentially has a ticket to the brain and central nervous system. Infections of the brain and nervous system do happen in influenza, but they are usually associated with "bird" flu strains.
A few years ago, Heaton's group published a paper that said some cells become infected by influenza and don't die. "There's this whole group of cells that get infected but do something different," he said. "So we felt like we should be studying those cells."
Heaton said his team wanted to focus on the body's first contact with the virus in the sinuses and upper respiratory tract to see if there were differences in immune responses from one cell to another. They found that the olfactory cells did become infected, but were able to fight off the virus and avoid being destroyed by it.
Key to the findings were the ability sort cells by type and then perform RNA sequencing that shows which genes are active in a given cell type. This allowed them to see that the olfactory cells were activating a known set of genes to respond to the invader, but turning them up much higher (or much faster, they aren't sure) than other cell types do."They upregulate genes that shut down viral replication," said Rebekah Dumm, a recent Ph.D. graduate of Duke who worked on this project as part of her dissertation. This is the same set of antiviral genes used by the epithelial cells, "but the higher level to which they are induced was striking," Dumm said. "It's surprising, because most of these genes were discovered and best described in the respiratory cells."
"Olfactory neurons have the ability to fight this virus off a little better than other cells," Heaton said. "We think that might be a brain protective mechanism."
Heaton said it would be fair to ask why all cells don't respond to influenza this way, but there is probably some tradeoff that isn't obvious yet. For example, a robust immune response that interfered with air exchange wouldn't be a winning strategy.
Heaton said his future work will continue to explore the immune response differences between cells. "You're a person who's been exposed to all kinds of stuff, you have your own particular exposure history that gives you different antibodies, different cell memory phenotypes, probably also different epithelial phenotypes, and we'd like to know what that means in terms of how you respond to secondary infections or these types of things."
More information: "Heterogeneity of Antiviral Responses in the Upper Respiratory Tract Mediates Differential Non-Lytic Clearance of Influenza Viruses," Rebekah Dumm, Sebastian Wellford, E. Ashley Moseman and Nicholas Heaton. Cell Reports, Sept. 1, 2020. DOI: 10.1016/j.celrep.2020.108103