New research supports sofosbuvir in combination with other antivirals for COVID-19
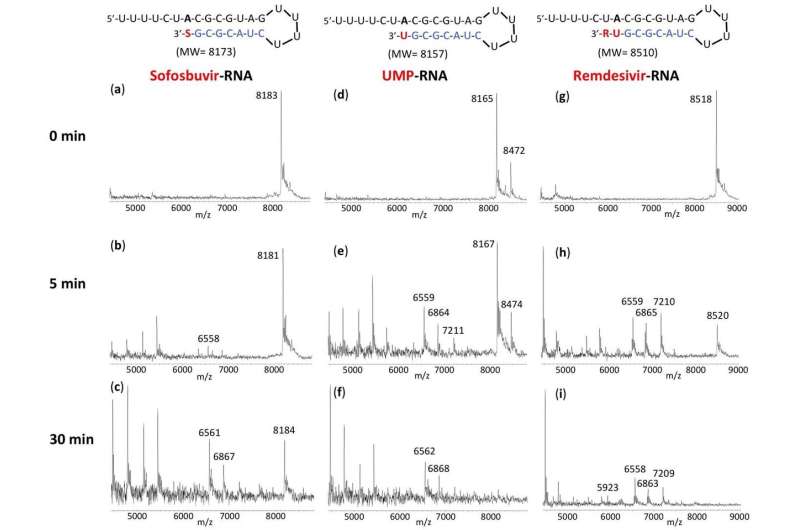
Columbia Engineering researchers report that Sofosbuvir-terminated RNA is more resistant to the proofreader of SARS-CoV-2, the virus that causes COVID-19, than Remdesivir-terminated RNA. The results of the new study, published today by the Nature Research journal Scientific Reports, support the use of the FDA-approved hepatitis C drug EPCLUSA—Sofosbuvir/Velpatasvir—in combination with other drugs in COVID-19 clinical trials.
The SARS-CoV-2 exonuclease-based proofreader maintains the accuracy of viral RNA genome replication to sustain virulence. Any effective antiviral targeting the SARS-CoV-2 polymerase must therefore display a certain level of resistance to this proofreading activity.
"We found that the RNA terminated by Sofosbuvir resists removal by the exonuclease to a substantially higher extent than RNA terminated by Remdesivir, another drug being used as a COVID-19 therapeutic," says the team's lead PI Jingyue Ju, Samuel Ruben-Peter G. Viele Professor of Engineering; professor of Chemical Engineering and Pharmacology; director, Center for Genome Technology & Biomolecular Engineering.
The new study builds upon earlier work the researchers have conducted. Last January, before COVID-19 reached pandemic status, the team posited that EPCLUSA might inhibit SARS-CoV-2, the virus responsible for COVID-19. Their reasoning was based on the analysis of the molecular structures and activities of hepatitis C viral inhibitors and a comparison of hepatitis C virus and coronavirus replication.
In a subsequent study, the researchers demonstrated that the active drug Sofosbuvir triphosphate is incorporated by SARS-CoV and SARS-CoV-2 polymerases, shutting down the polymerase reaction. Other investigators have since demonstrated the ability of Sofosbuvir to inhibit SARS-CoV-2 replication in lung and brain cells; currently, COVID-19 clinical trials with a number of hepatitis C drugs such as EPCLUSA and the combination of Sofosbuvir and Daclatasvir (which is similar to Velpatasvir) are ongoing in several countries.
Ju notes that a recent preprint from UC Berkeley indicates that a combination of Remdesivir and EPCLUSA increases Remdesivir's efficacy 25-fold in inhibiting SARS-CoV-2, the virus that causes COVID-19: "These results offer a molecular basis supporting the study of EPCLUSA in combination with Remdesivir for COVID-19 clinical trials."
More information: Steffen Jockusch et al, Sofosbuvir terminated RNA is more resistant to SARS-CoV-2 proofreader than RNA terminated by Remdesivir, Scientific Reports (2020). DOI: 10.1038/s41598-020-73641-9