EU regulator could approve Covid vaccines 'late 2020, early 2021'
Europe's medicines regulator said Monday that it could approve the first coronavirus vaccines late this year or early next, as it evaluates the most promising candidates.
The clarification came after EU Commission president Ursula von der Leyen said last week that the watchdog could give the green light for vaccines tested by Pfizer-BioNTech and by Moderna before the end of December.
"It is difficult to predict timelines for the authorisation of vaccines precisely at this stage, as the data are still coming in and the rolling reviews are currently ongoing," the European Medicines Agency said in an emailed statement to AFP.
"Depending on how the evaluation progresses, EMA could indeed be in a position to conclude evaluation for the most advanced candidates towards the end of this year or the beginning of next."
The Amsterdam-based EMA is carrying out "rolling reviews" designed to speed up vaccine approval times on three candidates: Pfizer-BioNTech, Moderna, and Oxford University-AstraZeneca.
US giant Pfizer and Germany's BioNTech has already sent data to the EMA after the companies said clinical trials showed their experimental vaccine is 95 percent effective.
US-based Moderna meanwhile says its vaccine is 94.5 percent effective, and Oxford and AstraZeneca said Monday that theirs showed 70 percent effectiveness, rising to 90 percent depending on the type of dosage.
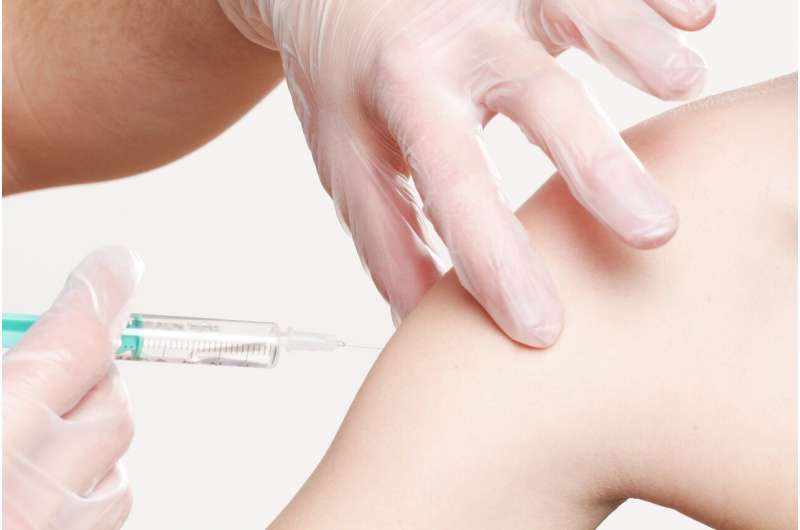
Several European countries have already unveiled strategies to roll out coronavirus vaccines to large parts of their populations as soon as they are approved by the EMA.
© 2020 AFP