November 27, 2020 feature
Immune responses during embryo development could increase risk of schizophrenia
Past research has often highlighted the effects that immune activation in pregnant women can have on the development of human embryos, for instance increasing the risk of a child developing psychiatric disorders later in life. The neural mechanisms underpinning these effects, however, remain largely unclear.
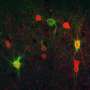
Researchers at New York Medical College have recently carried out a study investigating the impact that the activation of microglia (i.e., a specialized cell population that removes damaged neurons or infections) can have on an embryo's development of a specific class of neurons that regulates information processing, known as cortical interneurons. Their findings, published in Nature Neuroscience, suggest that activated microglia can cause metabolic disruptions that adversely impact the development of cortical interneurons. Interestingly, in individuals diagnosed with schizophrenia these disruptions could persist when the microglia are no longer activated.
"While we now known that cortical interneurons are affected by maternal immune activation, the mechanism through which they are affected is still poorly understood," Sangmi Chung, one of the researchers who carried out the study, told Medical Xpress. "Since the human embryo is not accessible for mechanistic studies, we used iPSC-derived human cortical interneurons to investigate how inflammation during development affects this vulnerable population of neurons."
Chung and her colleagues generated cortical interneurons using induced pluripotent stem cells (iPSCs), a technological tool that allows neuroscientists to reprogram cells that are extracted from human tissue samples. Past studies have found that individuals with schizophrenia present abnormal patterns in the functioning of cortical interneurons.
The researchers hoped that their study would enhance the present understanding of the neural mechanisms that may lead to the development of Schizophrenia or other neuropsychiatric disorders. The cortical interneurons used in their experiments were thus generated both from people with no psychiatric disorders and from patients diagnosed with schizophrenia.
After they created these cells, Chung and her colleagues co-cultured them either with or without activated microglial cells and observed the effects that these two procedures had on cortical interneurons generated from the tissue of healthy subjects and on those derived from that of patients with schizophrenia. Co-culture is a technique that enables the simultaneous cultivation of two or more different types of cells. The researchers specifically used a method called tissue culture insert, which allowed the signals released from microglia to reach the cortical interneurons they created through a membrane that was inserted between them, which is permeable but does not allow cells to pass through.
"We found that the metabolism of cortical interneurons is compromised under inflammatory condition during development, which showed prolonged impact in cortical interneurons derived from schizophrenia iPSCs but not healthy control iPSCs," Chung said. "Our findings highlight the existence of interactions between schizophrenia genetic backgrounds and environmental risk factors."
The findings gathered by Chung and her colleagues could inform future studies investigating the neural mechanisms that link prenatal immune activation with the risk of developing schizophrenia or other neuropsychiatric disorders. Most notably, it suggests that the activation of microglia, the cells that protect the nervous system against diseases and carry out immune responses, can cause metabolic disruptions in developmental cortical interneurons.
Moreover, the researchers found that in the cortical interneurons generated from the tissue of individuals with no neuropsychiatric disorders, these metabolic deficits were no longer present after the activated microglia were removed, while they persisted in patients affected by schizophrenia. Their findings could thus help to identify pre-natal neural processes that may interact with a person's genetic propensity for developing schizophrenia, increasing his/her risk of the developing the disorder later in life.
"We now plan to pursue further studies investigating the detailed mechanisms and pathways that are affected by inflammatory environments surrounding cortical interneurons," Chung said.
More information: Activated microglia cause metabolic disruptions in developmental cortical interneurons that persist in interneurons from individuals with schizophrenia. Nature Neuroscience(2020). DOI: 10.1038/s41593-020-00724-1
© 2020 Science X Network