South Africa considers reduced AstraZeneca rollout

South African health experts have suggested vaccinating several thousand people with AstraZeneca's COVID-19 vaccine to assess whether it protects against severe cases of a virus variant that emerged in the country.
Authorities delayed the start of a much-anticipated vaccination campaign—expected to begin this week with a first batch of one million AstraZeneca/Oxford doses—after a study showed the jabs failed to prevent mild and moderate infection from the South African variant.
The temporary suspension, announced late on Sunday, put a damper on the imminent rollout and stoked criticism of the coronavirus response in Africa's hardest-hit nation.
Top government advisor and leading epidemiologist Salim Abdool Karim said his team had suggested still using the AstraZeneca shots in a more staggered manner—starting with a smaller population group and closely studying their response.
"We would (first) vaccinate about 100,000 people or so to monitor hospitalisation (rates)," Karim told AFP on Monday, adding that the process would take "a few weeks".
"While we are rolling out the vaccine and collecting that information... we will be able to assess whether to continue."
Karim explained it was "prudent" to push back the wider distribution of AstraZeneca shots until scientists ascertain whether they could still prevent severe disease, hospitalisation and death.
He assured delay would be "partially compensated" by another batch of vaccines developed by US drugmaker Johnson & Johnson expected to land next week.
Vaccine 'portfolio'
The proposal will be fine-tuned with government over the coming days.
South Africa's Health Minister Zweli Mkhize told local media on Monday that an "implementation study" of AstraZeneca would take place alongside the rollout of Johnson & Johnson jabs.
Tests will be done "on the people that are getting the (AstraZeneca) vaccination," Mkhize said on SAFM radio.
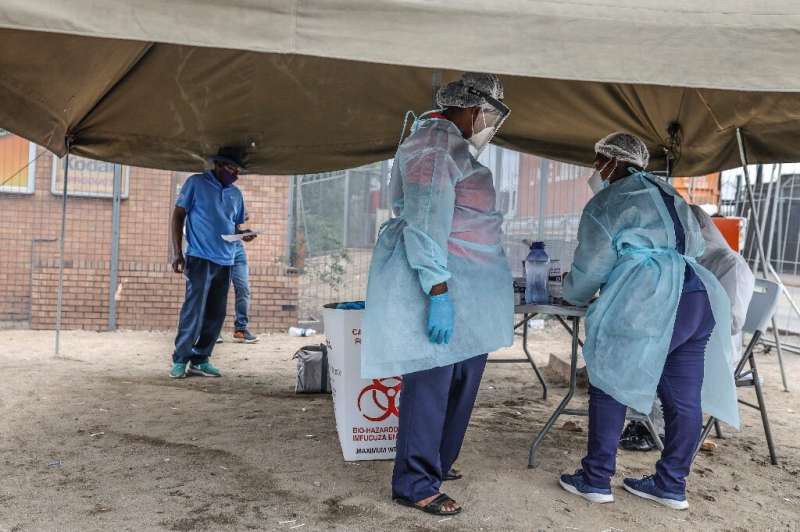
South Africa has lagged behind the global vaccination race and only received its first vaccines on February 1.
The one million AstraZeneca shots were produced by the Serum Institute of India, from which an additional 500,000 doses are expected this month.
The country aims to inoculate at least 67 percent of its population by the end of the year, or around 40 million people.
Some 1.2 million health workers will be first in line.
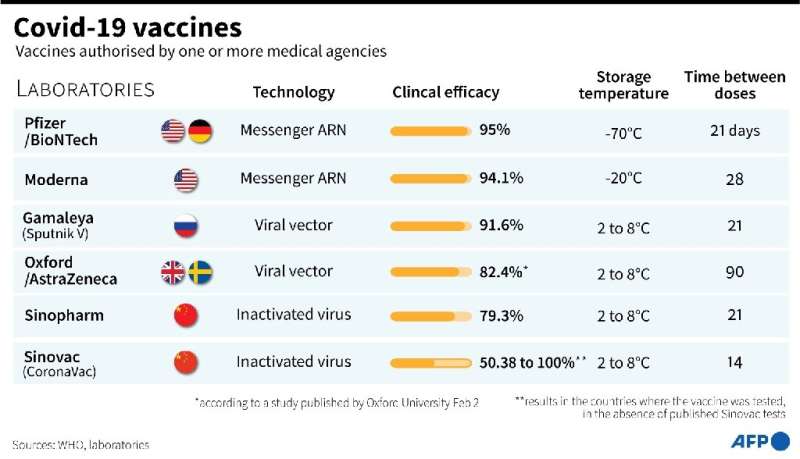
In addition to AstraZeneca and Johnson & Johnson, vaccines have been secured from US drugmaker Pfizer, the WHO-backed Covax facility and the African Union.

Discussions with other producers are also ongoing, particularly Moderna, Chinese manufacturers and the makers of the Russian Sputnik V jab.
South Africa's "vaccine strategy ensures that we are going to use a diverse portfolio of vaccines," said Karim.
"It so happens that we received the AstraZeneca vaccine first."
But the Democratic Alliance main opposition party accused government of lacking foresight.
"We should have taken into account the possibility of the changing efficacy or delays in rollout strategy," it said in a statement.
Rethinking priorities
The suspension of South Africa's vaccine rollout followed results of an AstraZeneca trial with 2,000 participants and which has not yet undergone peer review.
Johannesburg's University of Witwatersrand (Wits), which conducted the study, found the vaccine only "provides minimal protection against mild-moderate COVID-19 infection" from the South African variant.
Scientists believe that variant is more transmissible than its original form, fuelling an unprecedented spike in cases earlier this year.
Wits vaccinologist Clare Cutland noted the study's sample, with a median age of 31 and no co-morbidities, was too limited to assess AstraZeneca's efficacy against severe disease.
But she said a larger trial of the Johnson & Johnson vaccine, which uses similar technologies, showed relatively good protection against severe disease caused by the South African variant—suggesting there was a "good chance" the same would be observed with AstraZeneca.
"The results... mean that we need to pause to reassess," Cutland told AFP.
"Do we want it to prevent mild and moderate disease only, or... reduce and prevent severe disease?"
"We may not have herd immunity to protect against mild disease," she added. "And maybe that's not a huge issue."
South Africa has recorded almost 1.5 million coronavirus cases and over 46,200 deaths.
The mortality rate has remained relatively low, however, with at least 92 percent of cases recovered.
"The fact that this virus has mutated faster than we have changed the vaccine to catch up is unfortunate," said Cutland.
"The great thing is that the technologies are good... so the rollout of vaccines against different variants could be a lot quicker."
Researchers at AstraZeneca are working to update the vaccine and expect to have a version tailored to the South African variant by autumn.
© 2021 AFP