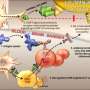
Researchers identify a novel autoantigen in narcolepsy, a mimic of a protein from swine flu virus
Researchers at the University of Helsinki have identified influenza virus peptides that resemble human protein fragments and can cause an immune response against the body's own cells due to cross-reactivity. The recently completed study confirms the notion that influenza A (H1N1) virus peptides can trigger an autoimmune reaction in genetically predisposed individuals, that results in the onset of narcolepsy with cataplexy.
Narcolepsy with cataplexy, or narcolepsy type 1 (NT1), is a rare and chronic neurological disease whose prevalence increased in children and adolescents after the administration of Pandemrix swine flu vaccine in 2009–2010. It is an autoimmune disease to which a specific inherited tissue type (HLA-DQB1*0602) predisposes people.
The disease mechanism of NT1 was investigated in a collaborative study carried out by Ph.D. student Arja Vuorela and university researcher Dr. Tobias Freitag, working in the research groups of Prof. Outi Vaarala and Prof. Seppo Meri. The study analyzed the cell-mediated immune response targeting three different proteins of influenza A (H1N1) virus included in the Pandemrix vaccine, in Finnish children and adolescents who developed narcolepsy after Pandemrix vaccination.
The study was published in the journal Nature Communications.
Immune cells are activated by the body's own proteins in narcolepsy patients
The researchers found that immune cells in the blood of narcolepsy patients mounted stronger responses against two peptides originating from swine flu virus neuraminidase or nucleoprotein, compared to the cells of vaccinated control subjects.
By investigating similarities between the two identified viral peptides and human proteins, the researchers identified similar sequences in two self proteins present in the human brain. This led them to study immune responses in narcolepsy patients also to these self proteins.
What they found demonstrated that patient lymphocytes recognised a fragment derived from the human protein-O-mannosyl-transferase 1 enzyme (POMT1). This POMT1 peptide mimics the peptide from influenza A (H1N1) neuraminidase, that is a target of the immune response generated by Pandemrix vaccine.
"Interestingly, immune cells in patient blood produced the same mediators of inflammation as a response to the peptides of both viral neuraminidase or human POMT1 enzymes. Both peptides also brought about the development and selection of identical or similar T lymphocyte clones," says University Researcher Tobias Freitag.
Onset of autoimmune diseases likely explained by cross-reactivity
Prior research has uncovered that "molecular mimicry," the similarity between proteins produced by pathogens or expressed in human tissues, may trigger the development of autoimmune diseases in people genetically at risk.
According to the latest study, lymphocytes which recognize the virus cross-react with human brain tissue and, consequently, contribute to the development of NT1. Autoantibodies recognizing the human POMT1 enzyme were also found at higher levels in individuals vaccinated with Pandemrix.
"The results indicate that the Pandemrix vaccine triggered an autoimmune response in genetically predisposed people. All of the individuals who developed narcolepsy had the same HLA-DQB1*0602 tissue type, which predisposes people to the disease. Most likely, the effective adjuvant included in the vaccine also contributed to the autoimmune response," says Professor of Immunology Seppo Meri from the University of Helsinki. NT1 is a multifactorial disease.
The findings can boost further research aimed at the improvement of diagnostics and treatments for NT1, including therapeutic strategies modifying or redirecting the immune system.
"The findings of this study serve as a reminder that no medical procedure, including vaccination, is entirely without risk. As with all clinical decision making, vaccination recommendations need to consider both benefits and possible adverse effects. When new vaccines are introduced, especially those based on novel adjuvants or vaccine technologies, recommendations need to be tailored to different age and risk groups," Freitag says.
More information: A. Vuorela et al. Enhanced influenza A H1N1 T cell epitope recognition and cross-reactivity to protein-O-mannosyltransferase 1 in Pandemrix-associated narcolepsy type 1, Nature Communications (2021). DOI: 10.1038/s41467-021-22637-8