Scientists identify 160 new drugs that could be repurposed against COVID-19
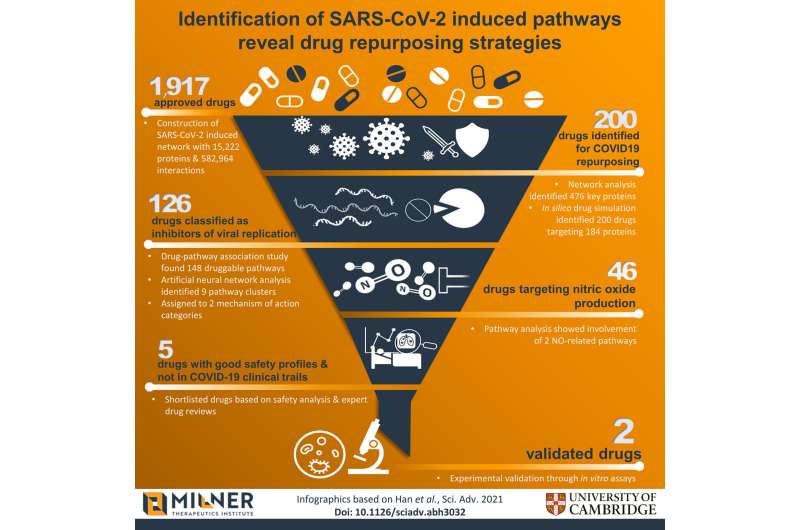
Cambridge scientists have identified 200 approved drugs predicted to work against COVID-19—of which only 40 are currently being tested in COVID-19 clinical trials.
In a study published today in Science Advances, a team led by researchers at the University of Cambridge's Milner Therapeutics Institute and Gurdon Institute used a combination of computational biology and machine learning to create a comprehensive map of proteins that are involved in SARS-CoV-2 infection—from proteins that help the virus break into the host cell to those generated as a consequence of infection. By examining this network using artificial intelligence (AI) approaches, they were able to identify key proteins involved in infection as well as biological pathways that might be targeted by drugs.
To date, the majority of small molecule and antibody approaches for treating COVID-19 are drugs that are either currently the subject of clinical trials or have already been through clinical trials and been approved. Much of the focus has been on several key virus or host targets, or on pathways—such as inflammation—where a drug treatment could be used as an intervention.
The team used computer modeling to carry out a 'virtual screen' of almost 2,000 approved drugs and identified 200 approved drugs that could be effective against COVID-19. Forty of these drugs have already entered clinical trials, which the researchers argue supports the approach they have taken.
When the researchers tested a subset of those drugs implicated in viral replication, they found that two in particular—an antimalarial drug and a type of medicine used to treat rheumatoid arthritis—were able to inhibit the virus, providing initial validation of their data-driven approach.
Professor Tony Kouzarides, Director of the Milner Therapeutics Institute, who led the study, said: "By looking across the board at the thousands of proteins that play some role in SARS-CoV-2 infection—whether actively or as a consequence of infections—we've been able to create a network uncovering the relationship between these proteins.
"We then used the latest machine learning and computer modeling techniques to identify 200 approved drugs that might help us treat COVID-19. Of these, 160 had not been linked to this infection before. This could give us many more weapons in our armory to fight back against the virus."
Using artificial neural network analysis, the team classified the drugs depending on the overarching role of their targets in SARS-CoV-2 infection: those that targeted viral replication and those that targeted the immune response. They then took a subset of those involved in viral replication and tested them using cell lines derived from humans and from non-human primates.
Of particular note were two drugs, sulfasalazine (used to treat conditions such as rheumatoid arthritis and Crohn's disease) and proguanil (and antimalarial drug), which the team showed reduced SARS-CoV-2 viral replication in cells, raising the possibility of their potential use to prevent infection or to treat COVID-19.
Dr. Namshik Han, Head of Computational Research and AI at the Milner Therapeutics Institute, added: "Our study has provided us with unexpected information about the mechanisms underlying COVID-19 and has provided us with some promising drugs that might be repurposed for either treating or preventing infection. While we took a data-driven approach—essentially allowing artificially intelligent algorithms to interrogate datasets—we then validated our findings in the laboratory, confirming the power of our approach.
"We hope this resource of potential drugs will accelerate the development of new drugs against COVID-19. We believe our approach will be useful for responding rapidly to new variants of SARS-CoV2 and other new pathogens that could drive future pandemics."
More information: N. Han el al., "Identification of SARS-CoV-2–induced pathways reveals drug repurposing strategies," Science Advances (2021). advances.sciencemag.org/lookup … .1126/sciadv.abh3032