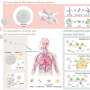
Nanocleaners penetrate the blood-brain barrier to clean toxic proteins and regulate inflammation in Alzheimer's disease
Insurmountable blood‒brain barrier (BBB) and complex pathological features are the key factors affecting the treatment of Alzheimer's disease (AD). Poor accumulation of drugs in lesion sites and undesired effectiveness of simply reducing Aβ deposition or TAU protein need to be resolved urgently.
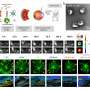
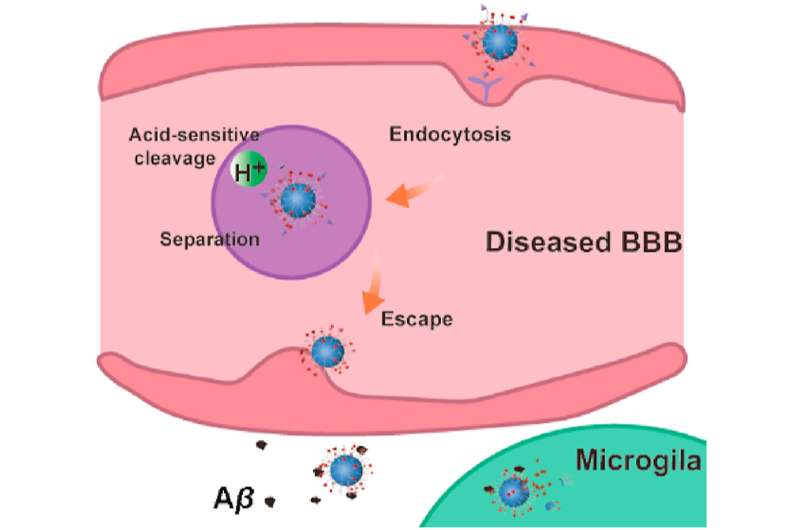
The authors of an article published in Acta Pharmaceutica Sinica B designed a nanocleaner with a rapamycin‒loaded ROS‒responsive PLGA core and surface modification with KLVFF peptide and acid‒cleavable DAG peptide (R@(ox‒PLGA)‒KcD). DAG can enhance the targeting and internalization effect of nanocleaner towards neurovascular unit endothelial cells in AD lesions, and subsequently detach from nanocleaner in response to acidic microenvironment of endosomes to promote the transcytosis of nanocleaner from endothelial cells into brain parenchyma. Then exposed KLVFF can capture and carry Aβ to microglia, attenuating Aβ‒induced neurotoxicity. Rapamycin, an autophagy promoter, is rapidly liberated from nanocleaner in the high ROS level of lesions to improve Aβ degradation and normalize inflammatory condition. This design altogether accelerates Aβ degradation and alleviates oxidative stress and excessive inflammatory response.
Collectively, the authors findings offer a strategy to target the AD lesions precisely and multi‒pronged therapies for clearing the toxic proteins and modulating lesion microenvironment, to achieve efficient AD therapy.
More information: Ting Lei et al, A nanocleaner specifically penetrates the blood‒brain barrier at lesions to clean toxic proteins and regulate inflammation in Alzheimer's disease, Acta Pharmaceutica Sinica B (2021). DOI: 10.1016/j.apsb.2021.04.022