Scientists discover novel mechanism of CircRNA inhibiting gastric cancer metastasis
Recently, researchers led by Prof. Lin Wenchu from the High Magnetic Field Laboratory, Hefei Institutes of Physical Science (HFIPS), Chinese Academy of Sciences (CAS), in collaboration with Prof. Shan Ge and Associate Prof. Hu Shanshan from the University of Science and Technology of China, proposed an elegant mechanism that one circRNA, called circURI1, exerted an anti-metastatic effect through sequestering hnRNPM to modulate alternative splicing in Gastric cancer (GC). This is the first reported association between circRNA and alternative splicing. The work was published in PNAS.
GC is the fifth most common malignant tumor and the third leading cause of cancer death worldwide, largely due to the presence of metastatic spread in patients. Circular RNAs (circRNAs) are a class of natural covalent closed single-stranded RNA molecules, which have emerged as key regulators of human cancers, yet their modes of action in GC remain obscure.
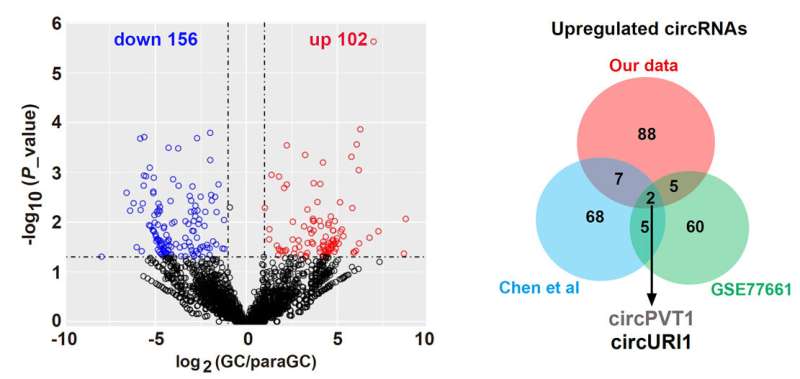
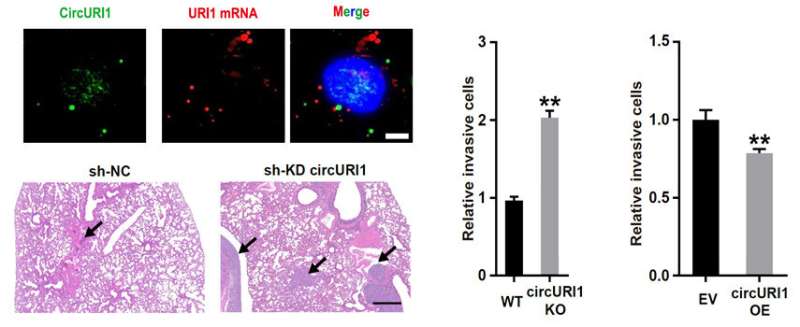
The researchers performed GC circRNA profiling and identified circURI1 with higher expressions in GC compared to the corresponding non-tumor tissues. Loss-of-function and gain-of-function studies revealed that circURI1 repressed cell migration and invasion in vitro and GC metastasis in vivo.
Mechanistically, circURI1 behaved as a decoy of hnRNPM in a sequence-dependent manner to modulate alternative splicing of a subset of genes related to cell migration, thus suppressing GC metastasis.
This study provides a new perspective on the molecular basis of cancer metastasis by circRNAs, and suggests that circURI1 as a potential target for diagnosis and treatment in gastric cancer.
More information: Xiaolin Wang et al, CircURI1 interacts with hnRNPM to inhibit metastasis by modulating alternative splicing in gastric cancer, Proceedings of the National Academy of Sciences (2021). DOI: 10.1073/pnas.2012881118