Mitigating lung damage, mortality due to SARS-CoV-2
In a new paper, researchers at the University of Illinois Chicago report that a drug approved for treating patients with autoimmune disease helped to prevent lung damage and death in mice infected with the SARS-CoV-2 virus, which causes COVID-19 in humans.
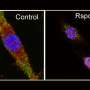
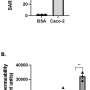
The results of their study provide strong evidence that inflammatory lung vascular leakage—or leaky lungs—is a key feature of COVID-19 illness. Vascular leakage can be caused by severe inflammation and results in a buildup of fluid in the lungs, which interferes with oxygen uptake. Mice infected with SARS-CoV-2 showed very clear and early signs of leakage from the blood vessels of the lung.
The research also suggests targeted drug treatments that suppress only select immune system pathways, like the rheumatoid arthritis drug used in the study that targets the molecular receptor called IL-1, might be a more suitable therapy for COVID-19 patients than drug treatments that suppress the entire immune system.
The study was led by senior author Asrar Malik, head of the department of pharmacology and regenerative medicine at the College of Medicine, and by co-senior author Jalees Rehman, professor of medicine in the department of pharmacology and regenerative medicine.
"With COVID-19, we need to strike a balance. On the one hand, we need a strong immune system to eliminate the virus. On the other hand, several studies suggest that in patients with severe COVID-19, the immune system can go overboard and even cause damage to our own body," Rehman said. "So, while we need the immune system to work efficiently, we also need to prevent it from becoming hyperactive and causing collateral damage."
The need for balance is why the UIC researchers decided to study the effects of a drug that works on only one targeted immune system pathway and see if that would help prevent SARS-CoV-2-induced leaky lungs.
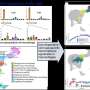
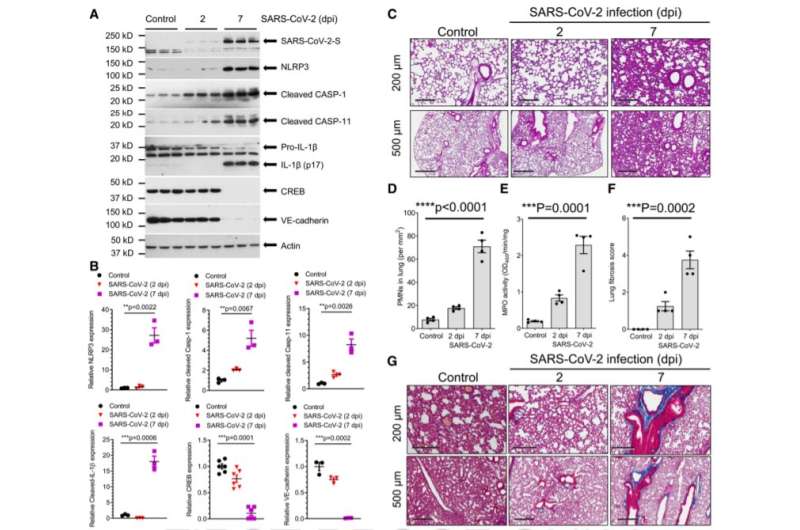
For the study, the researchers observed mice infected with the virus and tracked the progression of illness. They saw that the mice quickly showed symptoms like weight loss, fluid buildup in the lungs from leaky lung blood vessels, and even indicators of lung scarring, such as increased collagen levels in lung tissue.
"This is important evidence that blood vessel leakage in the lungs is a key feature of severe COVID-19 and that treatments which prevent or reduce vascular leakage warrant further study," Rehman said.
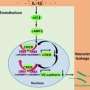
The researchers also treated some of the mice with the approved autoimmune disease drug, called anakinra, to block the IL-1 receptor, a key molecule regulating inflammation.
"We saw that the mice who received the drug had reduced signs of disease—including less lung fluid buildup and less scarring of the lungs—and better survival," Rehman said.
Rehman said these findings pave the way for helping COVID-19 patients and illuminate the need for more research on targeted, personalized treatments.
"Obviously, the best approach to reducing short-term and long-term damage as a result of COVID-19 is to get vaccinated and reduce the risk of SARS-CoV-2 infection as well as the risk of severe disease. However, the hesitancy of many individuals to get vaccinated as well as the lack of access to vaccines in many parts of the world means that we will continue to see patients with severe COVID-19 in the near future. Our results suggest that it is possible to identify a select a vulnerable COVID-19 patient population that is most likely to benefit from this therapy," Rehman said.
In their paper, "Interleukin-1RA Mitigates SARS-CoV-2–Induced Inflammatory Lung Vascular Leakage and Mortality in Humanized K18-hACE-2 Mice," the researchers hypothesize that by assessing the level of certain inflammatory signals in patients, such as the activation of the IL-1 receptor pathway, scientists could identify when a patient's immune system might be heading into overdrive and use a targeted immunosuppressant, like anakinra, to keep inflammation at the right balance.
"It is important to get the right drug to the right patient at the right time, and this study shines a light on a path forward for clinical trials that are investigating this drug and others that target specific components of the immune system," Rehman said.
More information: Shiqin Xiong et al, Interleukin-1RA Mitigates SARS-CoV-2–Induced Inflammatory Lung Vascular Leakage and Mortality in Humanized K18-hACE-2 Mice, Arteriosclerosis, Thrombosis, and Vascular Biology (2021). DOI: 10.1161/ATVBAHA.121.316925